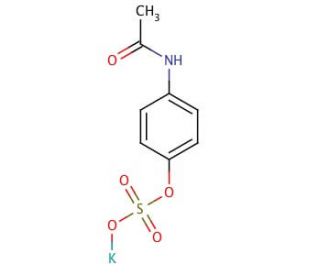
4-Acetaminophen Sulfate Potassium Salt (CAS 32113-41-0)
See product citations (5)
QUICK LINKS
4-Acetaminophen Sulfate Potassium Salt is a compound utilized in analytical chemistry for its role as a reference standard in the quantification of sulfate conjugates in various biological and environmental samples. It is an integral component in the development and validation of analytical methods, such as high-performance liquid chromatography (HPLC), which are essential for understanding sulfate conjugation pathways in metabolic studies. This salt form is specifically chosen for its increased solubility in aqueous solutions, enhancing its utility in laboratory assays. In biochemical research, it is used to investigate the mechanisms of sulfonation, a phase II metabolic reaction, and its impact on the bioavailability and metabolism of small organic molecules. Additionally, the potassium salt variant of 4-acetaminophen sulfate is applied in studies concerning the environmental fate of sulfated compounds, as it serves as a model for understanding how similar structures behave in natural water systems.
4-Acetaminophen Sulfate Potassium Salt (CAS 32113-41-0) References
- Carbonic anhydrase inhibitors. Inhibition of cytosolic isozymes I and II and transmembrane, tumor-associated isozyme IX with sulfamates including EMATE also acting as steroid sulfatase inhibitors. | Winum, JY., et al. 2003. J Med Chem. 46: 2197-204. PMID: 12747791
- Infrared studies on sulphate esters. III. O-Sulphate esters of alcohols, amino alcohols and hydroxylated amino acids. | LLOYD, AG., et al. 1961. Biochim Biophys Acta. 52: 413-9. PMID: 14466045
- Steroid sulphatase inhibitors for breast cancer therapy. | Purohit, A., et al. 2003. J Steroid Biochem Mol Biol. 86: 423-32. PMID: 14623540
- Simultaneous quantification of acetaminophen and structurally related compounds in human serum and plasma. | Bylda, C., et al. 2014. Drug Test Anal. 6: 451-60. PMID: 24000098
- Magnetic beads as an extraction medium for simultaneous quantification of acetaminophen and structurally related compounds in human serum. | Bylda, C., et al. 2015. Drug Test Anal. 7: 457-66. PMID: 25231125
- Time course of acetaminophen-protein adducts and acetaminophen metabolites in circulation of overdose patients and in HepaRG cells. | Xie, Y., et al. 2015. Xenobiotica. 45: 921-9. PMID: 25869248
- Simultaneous quantification of acetaminophen and five acetaminophen metabolites in human plasma and urine by high-performance liquid chromatography-electrospray ionization-tandem mass spectrometry: Method validation and application to a neonatal pharmacokinetic study. | Cook, SF., et al. 2015. J Chromatogr B Analyt Technol Biomed Life Sci. 1007: 30-42. PMID: 26571452
- Metabolism of AM404 From Acetaminophen at Human Therapeutic Dosages in the Rat Brain. | Muramatsu, S., et al. 2016. Anesth Pain Med. 6: e32873. PMID: 27110534
- The Impact of Proximal Roux-en-Y Gastric Bypass Surgery on Acetaminophen Absorption and Metabolism. | Chen, KF., et al. 2020. Pharmacotherapy. 40: 191-203. PMID: 31960977
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
4-Acetaminophen Sulfate Potassium Salt, 10 mg | sc-209906 | 10 mg | $516.00 | |||
4-Acetaminophen Sulfate Potassium Salt, 10 mg | sc-209906-CW | 10 mg | $669.00 |
