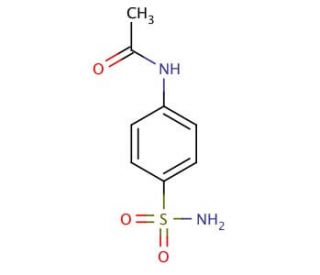
4-Acetamidobenzenesulfonamide (CAS 121-61-9)
QUICK LINKS
4-Acetamidobenzenesulfonamide functions as a carbonic anhydrase inhibitor. It acts by binding to the active site of the enzyme, preventing the conversion of carbon dioxide to bicarbonate and protons. This inhibition disrupts the normal physiological processes that rely on carbonic anhydrase activity, leading to downstream effects on pH regulation and ion transport. At the molecular level, 4-Acetamidobenzenesulfonamide forms specific interactions with amino acid residues within the active site of carbonic anhydrase, disrupting the enzyme′s catalytic function. 4-Acetamidobenzenesulfonamide′s mechanism of action involves interfering with the enzyme′s ability to facilitate biochemical reactions, ultimately impacting cellular processes that rely on carbonic anhydrase activity.
4-Acetamidobenzenesulfonamide (CAS 121-61-9) References
- Switching on prodrugs using radiotherapy. | Geng, J., et al. 2021. Nat Chem. 13: 805-810. PMID: 34112990
- Discovery of Novel 3-Phenylpiperidine Derivatives Targeting the β-Catenin/B-Cell Lymphoma 9 Interaction as a Single Agent and in Combination with the Anti-PD-1 Antibody for the Treatment of Colorectal Cancer. | Zhang, H., et al. 2023. J Med Chem. 66: 1349-1379. PMID: 36630177
- The design of small-molecule prodrugs and activatable phototherapeutics for cancer therapy. | Han, HH., et al. 2023. Chem Soc Rev. 52: 879-920. PMID: 36637396
- (η5-Cyclopentadienyl) Fe (CO) 2-complexes of sulfonamide anions. Acylation of the amino group in the sulfanilamide complex and its relevance to the protein labelling | Zakrzewski, J., & Pawlak, M. 1998. Journal of organometallic chemistry. 568(1-2): 171-175.
- Synthesis and antimicrobial evaluation of sulfanilamide-and carbohydrate-derived 1, 4-disubstitued-1, 2, 3-triazoles via click chemistry | Ay, K., Ispartaloğlu, B., Halay, E., Ay, E., Yaşa, İ., & Karayıldırım, T. 2017. Medicinal Chemistry Research. 26: 1497-1505.
- Two new diazonium bis (perfluoroalkyl) arylsulfonyl imide zwitterionic monomers from perfluoro (3-oxa-4-pentene) sulfonyl fluoride for proton exchange membrane fuel cells | Mei, H., & Ibrahim, F. 2017. Journal of Fluorine Chemistry. 199: 46-51.
- Efficient Approaches for the Synthesis of Diverse α-Diazo Amides. | Chow, Shiao, et al. 2020. Synthesis. 52.11: 1695-1706.
- Development of a Commercial Process for Deucravacitinib, a Deuterated API for TYK2 Inhibition | Treitler, D. S., Soumeillant, M. C., Simmons, E. M., Lin, D., Inankur, B., Rogers, A. J.,.. & Tymonko, S. 2022. Organic Process Research & Development. 26(4): 1202-1222.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
4-Acetamidobenzenesulfonamide, 25 g | sc-396804 | 25 g | $182.00 |
