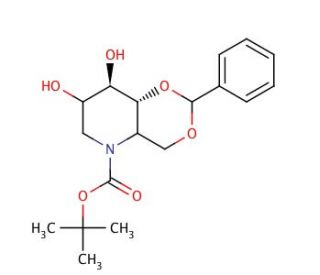
4,6-O-Benzylidene-N-(tert-butoxycarbonyl)-1,5-imino-D-glucitol (CAS 133697-16-2)
QUICK LINKS
4,6-O-Benzylidene-N-(tert-butoxycarbonyl)-1,5-imino-D-glucitol, a derivative of D-glucitol, holds significance in glycobiology research due to its structural resemblance to natural carbohydrate molecules and its versatile applications in studying glycan-mediated processes. Its mechanism of action primarily revolves around its role as a substrate in enzymatic glycosylation reactions. This compound serves as a glycosyl donor in the synthesis of glycoconjugates and glycosylated molecules, facilitating the exploration of carbohydrate-protein interactions and cellular recognition events. Moreover, 4,6-O-Benzylidene-N-(tert-butoxycarbonyl)-1,5-imino-D-glucitol serves as an essential intermediate in the synthesis of carbohydrate-based materials, including glycoarrays, carbohydrate-based sensors, and glycodendrimers. Its chemical reactivity allows for selective modifications, enabling the creation of tailored glycan structures for investigating glycan function and molecular recognition. Additionally, this compound finds applications in carbohydrate synthesis strategies for accessing diverse carbohydrate libraries, contributing to the development of novel glycan-based vaccines. Through its versatile applications, 4,6-O-Benzylidene-N-(tert-butoxycarbonyl)-1,5-imino-D-glucitol continues to advance our understanding of carbohydrate biology and pave the way for innovative research approaches aimed at unraveling the roles of glycans in various biological processes.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
4,6-O-Benzylidene-N-(tert-butoxycarbonyl)-1,5-imino-D-glucitol, 10 mg | sc-220996 | 10 mg | $380.00 |
