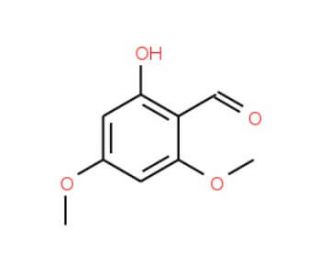
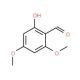
4,6-Dimethoxysalicylaldehyde (CAS 708-76-9)
QUICK LINKS
4,6-Dimethoxysalicylaldehyde is widely utilized in the synthesis of pharmaceuticals, dyes, and various organic compounds, showcasing its versatility. Moreover, it serves as a precursor in the synthesis of polymers and other compounds of interest. Additionally, it has played a pivotal role in the synthesis of peptides and proteins. One remarkable attribute of 4,6-Dimethoxysalicylaldehyde lies in its potent inhibition of the enzyme tyrosinase. Tyrosinase is responsible for the synthesis of melanin, the pigment found in the skin. Through its inhibitory action on tyrosinase, 4,6-Dimethoxysalicylaldehyde effectively reduces the production of melanin. Furthermore, the inhibitory effects of 4,6-Dimethoxysalicylaldehyde extend beyond tyrosinase. It has been observed to impede the growth of diverse fungi, bacteria, and even certain viruses. This property highlights its potential as useful in combating these harmful microorganisms.
4,6-Dimethoxysalicylaldehyde (CAS 708-76-9) References
- The first total synthesis of (+/-)-linderol A, a tricyclic hexahydrodibenzofuran constituent of Lindera umbellata bark, with potent inhibitory activity on melanin biosynthesis of cultured B-16 melanoma cells. | Yamashita, M., et al. 2001. Org Lett. 3: 1359-62. PMID: 11348234
- The ortho backbone amide linker (o-BAL) is an easily prepared and highly acid-labile handle for solid-phase synthesis. | Boas, U., et al. 2002. J Comb Chem. 4: 223-8. PMID: 12005482
- Investigations of intramolecular hydrogen bonding in three types of Schiff bases by 2H and 3H NMR isotope effects. | Schilf, W., et al. 2004. Magn Reson Chem. 42: 556-60. PMID: 15137048
- Antimicrobial properties of substituted salicylaldehydes and related compounds. | Pelttari, E., et al. 2007. Z Naturforsch C J Biosci. 62: 487-97. PMID: 17913062
- Synthesis and biodistribution of lipophilic and monocationic gallium radiopharmaceuticals derived from N,N'-bis(3-aminopropyl)-N,N'-dimethylethylenediamine: potential agents for PET myocardial imaging with 68Ga. | Hsiao, YM., et al. 2009. Nucl Med Biol. 36: 39-45. PMID: 19181267
- QSTR with extended topochemical atom (ETA) indices. 14. QSAR modeling of toxicity of aromatic aldehydes to Tetrahymena pyriformis. | Roy, K. and Das, RN. 2010. J Hazard Mater. 183: 913-22. PMID: 20739120
- {3,3',5,5'-Tetra-meth-oxy-2,2'-[ethane-1,2-diylbis(nitrilo-methyl-idyne)]diphenolato}nickel(II). | Assey, GE., et al. 2010. Acta Crystallogr Sect E Struct Rep Online. 66: m620. PMID: 21579278
- {3,3',5,5'-Tetra-meth-oxy-2,2'-[ethane-1,2-diylbis(nitrilo-methyl-idyne)]diphenol-ato}-copper(II). | Assey, G., et al. 2010. Acta Crystallogr Sect E Struct Rep Online. 66: m653. PMID: 21579299
- Bio- and chemical syntheses of mangiferin and congeners. | Ehianeta, TS., et al. 2016. Biofactors. 42: 445-458. PMID: 27774668
- Functional model for intradiol-cleaving catechol 1, 2-dioxygenase: Synthesis, structure, spectra, and catalytic activity of iron (III) complexes with substituted salicylaldimine ligands | Wang, C. H., Lu, J. W., Wei, H. H., & Takeda, M. 2007. Inorganica chimica acta. 360(9): 2944-2952.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
4,6-Dimethoxysalicylaldehyde, 1 g | sc-238993 | 1 g | $34.00 | |||
4,6-Dimethoxysalicylaldehyde, 5 g | sc-238993A | 5 g | $131.00 | |||
4,6-Dimethoxysalicylaldehyde, 10 g | sc-238993B | 10 g | $208.00 | |||
4,6-Dimethoxysalicylaldehyde, 25 g | sc-238993C | 25 g | $416.00 |
