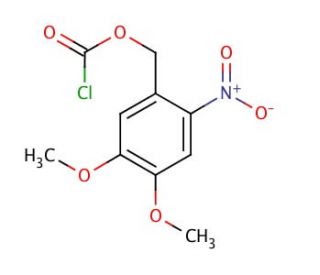
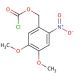
4,5-Dimethoxy-2-nitrobenzyl chloroformate (CAS 42855-00-5)
QUICK LINKS
4,5-Dimethoxy-2-nitrobenzyl chloroformate is a chemical reagent employed extensively in organic synthesis, particularly in the protection of amines. Its utility stems from its ability to form stable carbamates, which can be selectively deprotected under mild conditions. The compound is integral to research that involves peptide synthesis, as it offers a method for temporarily modifying the reactivity of amino groups. This reversible protection strategy allows for the sequential construction of complex peptide chains without unwanted side reactions. Moreover, its role in the study of protein structure and function is significant, as the protected intermediates can be used in various analytical techniques, including mass spectrometry and nuclear magnetic resonance spectroscopy, to probe the interactions within and between biomolecules. The compound is also a key player in the development of novel synthetic routes for the production of small organic molecules, which can include potential catalysts and ligands for use in further chemical transformations.
4,5-Dimethoxy-2-nitrobenzyl chloroformate (CAS 42855-00-5) References
- Hypoxia-selective nitrobenzyloxycarbonyl derivatives of 1,2-bis(methylsulfonyl)-1-(2-chloroethyl)hydrazines. | Shyam, K., et al. 1999. J Med Chem. 42: 941-6. PMID: 10072691
- Photochemical gating of heterologous ion channels: remote control over genetically designated populations of neurons. | Zemelman, BV., et al. 2003. Proc Natl Acad Sci U S A. 100: 1352-7. PMID: 12540832
- Encodamers: unnatural peptide oligomers encoded in RNA. | Frankel, A., et al. 2003. Chem Biol. 10: 1043-50. PMID: 14652071
- Development and pharmacological characterization of 'caged' urotensin II analogs. | Bourgault, S., et al. 2005. Peptides. 26: 1475-80. PMID: 16042988
- Caged capsaicins: New tools for the examination of TRPV1 channels in somatosensory neurons. | Gilbert, D., et al. 2007. Chembiochem. 8: 89-97. PMID: 17154194
- Branched intermediate formation stimulates peptide bond cleavage in protein splicing. | Frutos, S., et al. 2010. Nat Chem Biol. 6: 527-33. PMID: 20495572
- Light induced drug delivery into cancer cells. | Shamay, Y., et al. 2011. Biomaterials. 32: 1377-86. PMID: 21074848
- Photosensitive chitosan to control cell attachment. | Cheng, N. and Cao, X. 2011. J Colloid Interface Sci. 361: 71-8. PMID: 21679959
- Neuron-like PC12 cell patterning on a photoactive self-assembled monolayer. | Cheng, N. and Cao, X. 2013. J Biomed Mater Res A. 101: 3066-75. PMID: 23505148
- Design and application of light-activated probes for cellular signaling. | Chatenet, D., et al. 2015. Methods Mol Biol. 1234: 17-30. PMID: 25304345
- Statistical Methods for the Investigation of Solvolysis Mechanisms Illustrated by the Chlorides of the Carbomethoxy Protecting Groups NVOC and FMOC. | D'Souza, MJ., et al. 2015. J Anal Methods Chem. 2015: 941638. PMID: 25767738
- A high-throughput screening platform to identify nanocarriers for efficient delivery of RNA-based therapies. | Francisco, V., et al. 2021. Methods. 190: 13-25. PMID: 33359052
- Optical control of targeted protein degradation. | Reynders, M. and Trauner, D. 2021. Cell Chem Biol. 28: 969-986. PMID: 34115971
- Selective Deposition of Metals on Plastics Used in the Construction of Microanalytical Devices: Photo-Directed Formation of Metal Features on PMMA† | Alyssa C. Henry and Robin L. McCarley. 2001. J. Phys. Chem.B. 105: 8755–8761.
- A Dynamic Biomaterial Based on a 2-Nitrobenzyl Derivative with a tert-Butyl Substituent at the Benzyl Position: Rapid Response and Minimized Phototoxicity | Dr. Shota Yamamoto, Hiroki Ikegami, Prof. Kazuo Yamaguchi, Prof. Jun Nakanishi. 2018. 786-790. 2: 786-790.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
4,5-Dimethoxy-2-nitrobenzyl chloroformate, 1 g | sc-238989 | 1 g | $138.00 |
