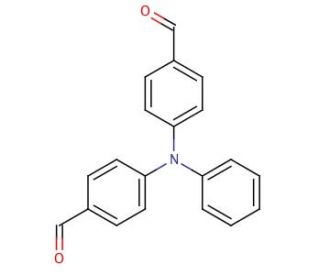
4,4′-Diformyltriphenylamine (CAS 53566-95-3)
QUICK LINKS
4,4′-Diformyltriphenylamine is a solid, crystalline substance that does not dissolve in water but is soluble in various organic solvents including chloroform, methanol, and ethanol. This compound is a building block in synthesizing materials with a diverse range of properties such as luminescence, electrochromicity, photosensitivity, and redox activity. 4,4′-Diformyltriphenylamine has good chemical stability, which makes it a suitable candidate for various applications that require stability under harsh chemical conditions. A purification process involving sublimation is used to rid the product of solvents and any undesirable impurities. It′s extensively used to fabricate fluorescent devices useful in analytical tools and lab experiments, including those measuring protein concentration. The fluorescence intensity this compound emits is correlated with the quantity of carbonyl groups in the molecule. Additionally, it′s instrumental in producing luminescence that can be quantified by devices equipped to identify fluorescence or phosphorescence. 4,4′-Diformyltriphenylamine exhibits maximum emission at a wavelength of 475 nm and is photochemically stable.
4,4′-Diformyltriphenylamine (CAS 53566-95-3) References
- Triphenylamine-Derived Solid-State Emissive Carbon Dots for Multicolor High-Efficiency Electroluminescent Light-Emitting Diodes. | Zhao, B., et al. 2023. Angew Chem Int Ed Engl. 62: e202301651. PMID: 36997339
- Hole Transport Triphenylamine−Azomethine Conjugated System: Synthesis and Optical, Photoluminescence, and Electrochemical Properties | Danuta Sek*†, Agnieszka Iwan†, Bozena Jarzabek†, Bozena Kaczmarczyk†, Janusz Kasperczyk†, Zbigniew Mazurak†, Marian Domanski†, Krzysztof Karon‡, and Mieczyslaw Lapkowski†‡. 2008. Macromolecules. 41: 6653–6663.
- Multifunctional, photochromic, acidichromic, electrochromic molecular switch: Novel aromatic poly(azomehine)s containing triphenylamine Group | Haijun Niu a b, Peihui Luo b, Milin Zhang a, Lin Zhang b, Lina Hao b, Jun Luo b, Xuduo Bai b, Wen Wang c. 2009. European Polymer Journal. 45: 3058-3071.
- Supramolecular Modification of Optical Properties of Some New Polyazomethines | Danuta Sek, Agnieszka Iwan, Bozena Kaczmarczyk & Bozena Jarzabek. 2010. Molecular Crystals and Liquid Crystals. 468: 119/[471]-129/[481].
- A study of thermal, optical and electrical properties of new branched triphenylamine-based polyazomethines | Danuta Sek a, Bozena Jarzabek a, Eugenia Grabiec a, Bozena Kaczmarczyk a, Henryk Janeczek a, Andrzej Sikora b, Agnieszka Hreniak b, Marcin Palewicz b, Mieczyslaw Lapkowski a c, Krzysztof Karon c, Agnieszka Iwan b. 2010. Synthetic Metals. 160: 2065-2076.
- Novel soluble polyazomethines with pendant carbazole and triphenylamine derivatives: preparation, characterization, and optical, electrochemical and electrochromic properties | Haijun Niu *ab, Hongqiang Kang ac, Jiwei Cai a, Cheng Wang a, Xuduo Bai *a and Wen Wang d. 2011. Polym. Chem. 2: 2804-2817.
- New naphthalene diimide-based compounds containing triarylamine units and imine linkages: Thermal, optical and electrochemical properties | Ewa Schab-Balcerzak a b, Marzena Grucela-Zajac a, Michal Krompiec a, Henryk Janeczek b, Mariola Siwy b, Danuta Sek b. 2011. Synthetic Metals. 161: 2268-2279.
- Opto(electrical) properties of new aromatic polyazomethines with fluorene moieties in the main chain for polymeric photovoltaic devices | Agnieszka Iwan, Marcin Palewicz, Andrzej Chuchmała, Lech Gorecki, Andrzej Sikora, Boleslaw Mazurek, Grzegorz Pasciak. 2012. Synthetic Metals. 162: 143-153.
- Tuning the bandgaps of polyazomethines containing triphenylamine by different linkage sites of dialdhyde monomers | Jiwei Cai a, Haijun Niu a b, Cheng Wang a, Lina Ma a, Xuduo Bai a, Wen Wang c. 2012. Electrochimica Acta. 76: 229-241.
- FTIR study of conjugation in selected aromatic polyazomethines | Bozena Kaczmarczyk. 2013. Journal of Molecular Structure. 1048: 179-184.
- Simple approach to regulate the spectra of novel kinds of polyazomethines containing bulky triphenylamine: Electrochemistry, electrochromism and photophysical responsive to environment | Haijun Niu a b, Jiwei Cai a, Ping Zhao b, Cheng Wang a, Xuduo Bai a, Wen Wang c. 2013. Dyes and Pigments. 96: 158-169.
- Red emitting solid state fluorescent triphenylamine dyes: Synthesis, photo-physical property and DFT study | Vinod D. Gupta, Abhinav B. Tathe, Vikas S. Padalkar, Prashant G. Umape, Nagaiyan Sekar. 2013. Dyes and Pigments. 97: 429-439.
- Electronic and thermal properties of compounds bearing diimide, azomethine and triphenylamine units | Marzena Grucela-Zajac a, Katarzyna Bijak a, Ernestas Zaleckas b, Saulius Grigalevicius b, Malgorzata Wiacek a, Henryk Janeczek c, Ewa Schab-Balcerzak a c. 2014. Optical Materials. 37: 543-551.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
4,4′-Diformyltriphenylamine, 1 g | sc-233036 | 1 g | $230.00 |
