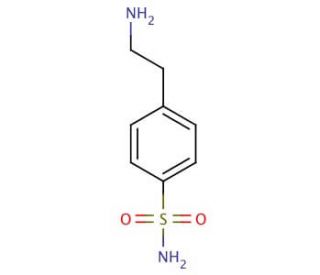
4-(2-Aminoethyl)benzenesulfonamide (CAS 35303-76-5)
QUICK LINKS
4-(2-Aminoethyl)benzenesulfonamide holds significance in scientific research with its diverse applications. This organic compound exists as a white, crystalline solid, readily soluble in water and ethanol. Its prominence stems from being a building block in synthetic organic chemistry, finding utility in synthesizing an array of compounds. The scientific research applications of 4-(2-Aminoethyl)benzenesulfonamide encompass its role in synthesizing dyes, and other organic compounds. Additionally, it serves as a foundational component in generating polymers, surfactants, and catalysts. The versatility of this compound extends to the synthesis of peptides, and proteins, making it useful in the field. The exact mechanism of action of 4-(2-Aminoethyl)benzenesulfonamide remains partially understood. Nevertheless, it is believed to function as an inhibitor of diverse enzymes, including proteases and kinases.
4-(2-Aminoethyl)benzenesulfonamide (CAS 35303-76-5) References
- Synthesis of Schiff base derivatives of 4-(2-aminoethyl)-benzenesulfonamide with inhibitory activity against carbonic anhydrase isoforms I, II, IX and XII. | Durgun, M., et al. 2015. Bioorg Med Chem Lett. 25: 2377-81. PMID: 25913118
- Synthesis and carbonic anhydrase inhibitory properties of novel 4-(2-aminoethyl)benzenesulfonamide-dipeptide conjugates. | Küçükbay, H., et al. 2019. Bioorg Chem. 83: 414-423. PMID: 30419497
- Enhanced nuclear gene delivery via integrating and streamlining intracellular pathway. | Qi, LY., et al. 2022. J Control Release. 341: 511-523. PMID: 34864117
- Macrophage-Mimic Hollow Mesoporous Fe-Based Nanocatalysts for Self-Amplified Chemodynamic Therapy and Metastasis Inhibition via Tumor Microenvironment Remodeling. | Zuo, W., et al. 2022. ACS Appl Mater Interfaces. 14: 5053-5065. PMID: 35040616
- Identification of Novel and Potent Indole-Based Benzenesulfonamides as Selective Human Carbonic Anhydrase II Inhibitors: Design, Synthesis, In Vitro, and In Silico Studies. | Elkamhawy, A., et al. 2022. Int J Mol Sci. 23: PMID: 35269684
- Squaramide-Tethered Sulfonamides and Coumarins: Synthesis, Inhibition of Tumor-Associated CAs IX and XII and Docking Simulations. | Arrighi, G., et al. 2022. Int J Mol Sci. 23: PMID: 35887037
- Design, Synthesis, Molecular Modeling and Anti-Hyperglycemic Evaluation of Quinazoline-Sulfonylurea Hybrids as Peroxisome Proliferator-Activated Receptor Gamma (PPARγ) and Sulfonylurea Receptor (SUR) Agonists. | El-Zahabi, MA., et al. 2022. Int J Mol Sci. 23: PMID: 36077003
- Identification of a glycolysis- and lactate-related gene signature for predicting prognosis, immune microenvironment, and drug candidates in colon adenocarcinoma. | Liu, C., et al. 2022. Front Cell Dev Biol. 10: 971992. PMID: 36081904
- Development of Hydrogen Sulfide-Releasing Carbonic Anhydrases IX- and XII-Selective Inhibitors with Enhanced Antihyperalgesic Action in a Rat Model of Arthritis. | Bonardi, A., et al. 2022. J Med Chem. 65: 13143-13157. PMID: 36121705
- Design and synthesis of benzothiazole-based SLC-0111 analogues as new inhibitors for the cancer-associated carbonic anhydrase isoforms IX and XII. | Al-Warhi, T., et al. 2022. J Enzyme Inhib Med Chem. 37: 2635-2643. PMID: 36146927
- Ratiometric Zinc Biosensor Based on Bioluminescence Resonance Energy Transfer: Trace Metal Ion Determination with Tunable Response. | Matveeva, EG., et al. 2022. Int J Mol Sci. 23: PMID: 36499262
- A Self-Disguised Nanospy for Improving Drug Delivery Efficiency via Decreasing Drug Protonation. | Zhang, K., et al. 2023. Small. 19: e2300060. PMID: 36929045
- Promising Schiff bases in antiviral drug design and discovery. | Kaushik, S., et al. 2023. Med Chem Res. 32: 1063-1076. PMID: 37305208
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
4-(2-Aminoethyl)benzenesulfonamide, 25 g | sc-232204 | 25 g | $42.00 |
