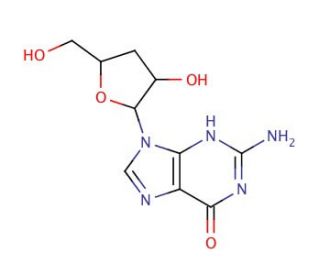
3′-Deoxyguanosine (CAS 3608-58-0)
See product citations (2)
QUICK LINKS
3′-Deoxyguanosine (3′-dG) is a naturally occurring modified form of guanosine, a purine nucleoside present in DNA and RNA. Extensively studied due to its potential applications across various fields, from biochemistry to medicine. It serves as a key component in the structure and function of DNA and RNA, making it an essential subject of investigation in scientific research. The mechanism of action of 3′-Deoxyguanosine is multifaceted and still not completely comprehended. However, it is understood that 3′-Deoxyguanosine participates in the formation and metabolism of DNA and RNA, highlighting its integral role in these processes. Furthermore, 3′-Deoxyguanosine is involved in protein metabolism and the formation of proteins.
3′-Deoxyguanosine (CAS 3608-58-0) References
- Acquisition of transcriptional competence in the 1-cell mouse embryo: requirement for recruitment of maternal mRNAs. | Aoki, F., et al. 2003. Mol Reprod Dev. 64: 270-4. PMID: 12548659
- Inhibition of the replication of a hepatitis C virus-like RNA template by interferon and 3'-deoxycytidine. | King, RW., et al. 2002. Antivir Chem Chemother. 13: 363-70. PMID: 12718408
- Rat brain guanosine binding site. Biological studies and pseudo-receptor construction. | Traversa, U., et al. 2003. Bioorg Med Chem. 11: 5417-25. PMID: 14642586
- Conformers of guanosines and their vibrations in the electronic ground and excited states, as revealed by double-resonance spectroscopy and ab initio calculations. | Nir, E., et al. 2004. Chemphyschem. 5: 131-7. PMID: 14999856
- Effects of polyadenylation inhibition on meiosis progression in relation to the polyadenylation status of cyclins A2 and B1 during in vitro maturation of bovine oocytes. | Traverso, JM., et al. 2005. Mol Reprod Dev. 71: 107-14. PMID: 15736128
- Structure of human PNP complexed with ligands. | Canduri, F., et al. 2005. Acta Crystallogr D Biol Crystallogr. 61: 856-62. PMID: 15983407
- Synthesis and antiviral evaluation of carbocyclic analogues of 2-amino-6-substituted-purine 3'-deoxyribofuranosides. | Shealy, YF., et al. 1987. J Med Chem. 30: 1090-4. PMID: 3035178
- The effects of purine nucleoside analogs on the response of the RIF-1 tumor to melphalan in vivo. | Horsman, MR., et al. 1986. Int J Radiat Oncol Biol Phys. 12: 801-6. PMID: 3486861
- The role in cancer therapy of inhibiting recovery from PLD induced by radiation or bleomycin. | Nakatsugawa, S. and Dewey, WC. 1984. Int J Radiat Oncol Biol Phys. 10: 1425-30. PMID: 6206038
- PLDR inhibitors: their biological and clinical implications. | Nakatsugawa, S., et al. 1984. Br J Cancer Suppl. 6: 43-7. PMID: 6607738
- Effects of inhibitors of radiation-induced potentially lethal damage repair on chemotherapy in murine tumors. | Nakatsugawa, S. and Sugahara, T. 1982. Int J Radiat Oncol Biol Phys. 8: 1555-9. PMID: 6982887
- 3'-Deoxyribonucleotides inhibit eukaryotic DNA primase. | Izuta, S., et al. 1996. J Biochem. 119: 1038-44. PMID: 8827435
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
3′-Deoxyguanosine, 10 mg | sc-220903 | 10 mg | $84.00 |
