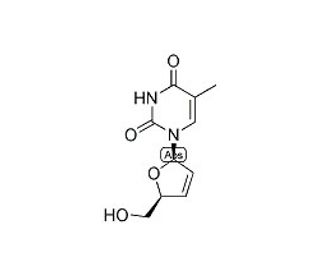
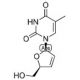
Molecular structure of 3′-Deoxy-2′,3′-didehydrothymidine, CAS Number: 3056-17-5
3′-Deoxy-2′,3′-didehydrothymidine (CAS 3056-17-5)
Alternate Names:
Stavudine; Sanilvudine; 2′,3′-Didehydro-2′,3′-dideoxythymidine; 3′-Deoxy-2′,3′-didehydro-D-thymidine; 2′,3′-Didehydro-3′-deoxythymidine
Application:
3′-Deoxy-2′,3′-didehydrothymidine is a HIV reverse transcriptase (HIV-1 RT) inhibitor
CAS Number:
3056-17-5
Purity:
≥98%
Molecular Weight:
224.2
Molecular Formula:
C10H12N2O4
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
3′-Deoxy-2′,3′-didehydrothymidine is a HIV-1 RT (HIV reverse transcriptase) inhibitor that causes a consistent and rapid shortening of telomeres in Tetrahymera. 3′-Deoxy-2′,3′-didehydrothymidine may act to decrease the proliferative activity of Jurkat and L1210 cell lines as well as human peripheral blood mononuclear cells at 250 muM concentration.
3′-Deoxy-2′,3′-didehydrothymidine (CAS 3056-17-5) References
- The effects of telomerase inhibitors on lymphocyte function. | Beltz, L., et al. 1999. Anticancer Res. 19: 3205-11. PMID: 10652613
- Substrate specificity of Escherichia coli thymidine phosphorylase for pyrimidine nucleosides with anti-human immunodeficiency virus activity. | Schinazi, RF., et al. 1992. Biochem Pharmacol. 44: 199-204. PMID: 1322661
- Synthesis of pyranoid analogues of the anti-HIV active 3'-deoxy-2',3'-didehydrothymidine (D4T). | Hansen, HB., et al. 1992. Arch Pharm (Weinheim). 325: 491-7. PMID: 1329694
- Identification and characterization of the conserved nucleoside-binding sites in the Epstein-Barr virus thymidine kinase. | Wu, CC., et al. 2004. Biochem J. 379: 795-803. PMID: 14705959
- Bis-ketol nucleoside triesters as prodrugs of the antiviral nucleoside triphosphate analogues of 3'-deoxythymidine and 3'-deoxy-2',3'-didehydrothymidine. | Calvo, KC., et al. 2004. Nucleosides Nucleotides Nucleic Acids. 23: 637-46. PMID: 15113029
- Pharmacokinetics of 3'-fluoro-3'-deoxythymidine and 3'-deoxy-2',3'-didehydrothymidine in rats. | Boudinot, FD., et al. 1991. Antimicrob Agents Chemother. 35: 747-9. PMID: 1648888
- [Isosteric triphosphonate analogues of dNTP: synthesis and substrate properties toward various DNA polymerases]. | Skoblov, AIu., et al. 2007. Bioorg Khim. 33: 527-37. PMID: 18050658
- Diastereoselective synthesis of aryloxy phosphoramidate prodrugs of 3'-deoxy-2',3'-didehydrothymidine monophosphate. | Roman, CA., et al. 2010. J Med Chem. 53: 7675-81. PMID: 20945915
- Pharmacokinetics of 3'-fluoro-3'-deoxythymidine and 3'-deoxy-2',3'-didehydrothymidine in rhesus monkeys. | Schinazi, RF., et al. 1990. Antimicrob Agents Chemother. 34: 1214-9. PMID: 2168144
- Comparison of metabolism and in vitro antiviral activity of stavudine versus other 2',3'-dideoxynucleoside analogues. | Sommadossi, JP. 1995. J Infect Dis. 171 Suppl 2: S88-92. PMID: 7861023
- Empirical and rational approaches for development of inhibitors of the human immunodeficiency virus--HIV-1. | Prusoff, W., et al. 1993. Pharmacol Ther. 60: 315-29. PMID: 8022862
- In vitro and in vivo disposition and metabolism of 3'-deoxy-2',3'-didehydrothymidine. | Cretton, EM., et al. 1993. Antimicrob Agents Chemother. 37: 1816-25. PMID: 8239589
- Cytotoxic interactions of 5-fluorouracil and nucleoside analogues in vitro. | Li, YX., et al. 1997. Anticancer Res. 17: 21-7. PMID: 9066626
Inhibitor of:
HIV-1 RT.Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
3′-Deoxy-2′,3′-didehydrothymidine, 25 mg | sc-202420 | 25 mg | $111.00 |
