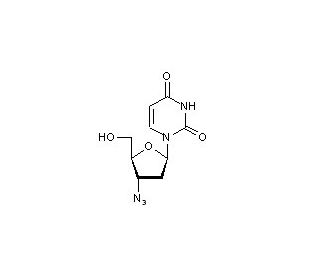
3′-Azido-2′,3′-dideoxyuridine (CAS 84472-85-5)
QUICK LINKS
3′-Azido-2′,3′-dideoxyuridine is a nucleoside analog that is structurally similar to thymidine, but with critical modifications that include the absence of a 3′-hydroxyl group on the sugar moiety and the substitution of the 3′-hydrogen with an azido (N3) group. These structural changes are fundamental to its action as it impacts the molecule′s interaction with nucleic acids and enzymes involved in nucleic acid synthesis. The primary mechanism of action of 3′-Azido-2′,3′-dideoxyuridine involves its incorporation into DNA during the replication process. Due to the presence of the azido group at the 3′ position, this compound acts as a chain terminator. Once incorporated into a growing DNA strand, it prevents the addition of further nucleotides by blocking the formation of a necessary 3′-5′ phosphodiester bond with the next nucleotide. This termination of DNA synthesis is crucial for studying the replication processes and the effects of modified nucleotides on DNA polymerase activity. In research, 3′-Azido-2′,3′-dideoxyuridine has been used extensively to investigate viral replication mechanisms, particularly for viruses that rely heavily on host cellular machinery to replicate their genomes. By incorporating into viral DNA, it allows researchers to halt replication at specific sites, helping to map where nucleoside analogs interfere with viral enzymes and to understand the broader implications of nucleotide modifications on viral life cycles. Furthermore, 3′-Azido-2′,3′-dideoxyuridine is utilized in studies focusing on the fidelity and mechanism of DNA polymerases. It provides insights into how DNA polymerases discriminate between natural nucleotides and modified versions, contributing to the understanding of enzyme substrate specificity and the potential for inducing mutagenesis through altered nucleotide incorporation.
3′-Azido-2′,3′-dideoxyuridine (CAS 84472-85-5) References
- Determination of 3'-azido-2',3'-dideoxyuridine in maternal plasma, amniotic fluid, fetal and placental tissues by high-performance liquid chromatography. | Clark, TN., et al. 2001. J Chromatogr B Biomed Sci Appl. 755: 165-72. PMID: 11393701
- Catabolic disposition of 3'-azido-2',3'-dideoxyuridine in hepatocytes with evidence of azido reduction being a general catabolic pathway of 3'-azido-2',3'-dideoxynucleosides. | Cretton, EM., et al. 1992. Biochem Pharmacol. 44: 973-80. PMID: 1326966
- Simultaneous determination of 3'-azido-2',3'-dideoxyuridine and novel prodrugs in rat plasma by liquid chromatography. | Kong, L., et al. 2003. J Chromatogr B Analyt Technol Biomed Life Sci. 795: 371-6. PMID: 14522042
- Pharmacokinetic evaluation of 3'-azido-2', 3'-dideoxyuridine-5'-O-valinate-hydrochloride as a prodrug of the anti-HIV nucleoside 3'-azido-2', 3'-dideoxyuridine. | Kong, L., et al. 2003. Antivir Chem Chemother. 14: 263-70. PMID: 14694989
- 3'-Azido-2',3'-dideoxyuridine (AzddU): comparative pharmacokinetics with 3'-azido-3'-deoxythymidine (AZT) in monkeys. | Boudinot, FD., et al. 1990. AIDS Res Hum Retroviruses. 6: 219-28. PMID: 2328158
- Phosphorylation of 3'-azido-2',3'-dideoxyuridine and preferential inhibition of human and simian immunodeficiency virus reverse transcriptases by its 5'-triphosphate. | Eriksson, BF., et al. 1989. Antimicrob Agents Chemother. 33: 1729-34. PMID: 2480079
- Comparative pharmacokinetics of 3'-azido-3'-deoxythymidine (AZT) and 3'-azido-2',3'-dideoxyuridine (AZddU) in mice. | Doshi, KJ., et al. 1989. Drug Metab Dispos. 17: 590-4. PMID: 2575492
- Synthesis and antiviral activity of several 2,5'-anhydro analogues of 3'-azido-3'-deoxythymidine, 3'-azido-2',3'-dideoxyuridine, 3'-azido-2',3'-dideoxy-5-halouridines, and 3'-deoxythymidine against human immunodeficiency virus and Rauscher-murine leukemia virus. | Lin, TS., et al. 1989. J Med Chem. 32: 1891-5. PMID: 2754712
- Interaction of the 5'-phosphates of the anti-HIV agents, 3'-azido-3'-deoxythymidine and 3'-azido-2',3'-dideoxyuridine, with thymidylate synthase. | Dzik, JM., et al. 1988. Biochem Biophys Res Commun. 155: 1418-23. PMID: 3178819
- Lymphatic distribution of 3'-azido-3'-deoxythymidine and 3'-azido-2',3'-dideoxyuridine in mice. | Manouilov, KK., et al. 1995. Drug Metab Dispos. 23: 655-8. PMID: 7587948
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
3'-Azido-2',3'-dideoxyuridine, 10 mg | sc-216614 | 10 mg | $300.00 |
