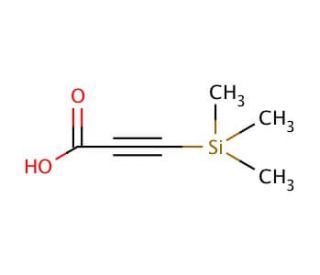
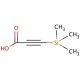
3-(Trimethylsilyl)propynoic acid (CAS 5683-31-8)
QUICK LINKS
3-(Trimethylsilyl)propiolic acid, referred to as 3-TSP, serves as a versatile and potent reagent with a multitude of applications in the realm of organic synthesis. This silylating agent finds use in diverse reactions, including the creation of various compounds and safeguarding functional groups. Moreover, it plays a role in preparing organometallic compounds, chiral compounds with high enantiomeric purity, and polymers, rendering it indispensable in biotechnology industries. In the realm of scientific research, 3-(Trimethylsilyl)propiolic acid extends its applications even further. It plays a pivotal role in the synthesis of organometallic compounds, chiral compounds with precise enantiomeric properties, and polymers. Additionally, its versatility allows for the synthesis of a wide array of organic compounds, such as peptides, carbohydrates, nucleic acids, and polysaccharides. The compound also finds utility in protecting functional groups like amines, alcohols, carboxylic acids, and thiols. The reaction of 3-(Trimethylsilyl)propiolic acid with a variety of substrates, including alcohols, amines, carboxylic acids, and thiols, follows a well-established process. It involves the formation of a silyl ether, which is subsequently hydrolyzed to produce the desired end product. For catalyzing this reaction, bases like potassium hydroxide or sodium hydroxide are employed. Overall, 3-(Trimethylsilyl)propiolic acid stands as a and adaptable compound, showcasing its significant impact in various organic syntheses and scientific advancements.
3-(Trimethylsilyl)propynoic acid (CAS 5683-31-8) References
- Complete 1H and 13C spectral assignment of floridoside. | Simon-Colin, C., et al. 2002. Carbohydr Res. 337: 279-80. PMID: 11844498
- Total syntheses of the coumarin-containing natural products pimpinellin and fraxetin using Au(I)-catalyzed intramolecular hydroarylation (IMHA) chemistry. | Cervi, A., et al. 2013. J Org Chem. 78: 9876-82. PMID: 23977955
- Butyrolactone synthesis via polar radical crossover cycloaddition reactions: diastereoselective syntheses of methylenolactocin and protolichesterinic acid. | Zeller, MA., et al. 2014. Org Lett. 16: 4810-3. PMID: 25190259
- Synthesis of carboxy-polyethylene glycol-amine (CA (PEG)n) and [1-14 C]-CA (PEG)n via oxa-Michael addition of amino-polyethylene glycols to propiolates vs to acrylates. | Song, F., et al. 2020. J Labelled Comp Radiopharm. 63: 15-24. PMID: 31736118
- Copper-Catalyzed Decarboxylative Cycloaddition of Propiolic Acids, Azides, and Arylboronic Acids: Construction of Fully Substituted 1,2,3-Triazoles. | Wang, XX., et al. 2020. J Org Chem. 85: 3576-3586. PMID: 31984747
- NMR internal standard signal shifts due to cyclodextrin inclusion complexes. | Ray, CL., et al. 2022. Magn Reson Chem. 60: 80-85. PMID: 33963614
- Palladium and copper catalyzed Sonogashira decarboxylative coupling of aryl iodides and alkynyl carboxylic acids | Carine Maaliki, Yoan Chevalier, Emilie Thiery, Jérôme Thibonnet. 2016. Tetrahedron Letters. 57: 3358-3362.
- Ag2CO3-catalyzed efficient synthesis of internal or terminal propargylicamines and chalcones via A3-coupling under solvent-free condition | Ningbo Li a c 1, Shitang Xu b 1, Xueyan Wang b, Li Xu a, Jie Qiao a c, Zhiwu Liang b, Xinhua Xu b. 2021. Chinese Chemical Letters. 32: 3993-3997.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
3-(Trimethylsilyl)propynoic acid, 1 g | sc-238482 | 1 g | $72.00 |
