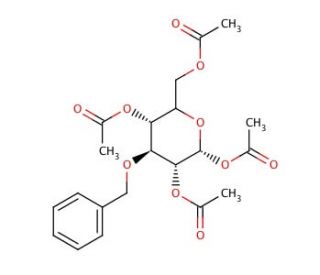
3-O-Benzyl-1,2,4,6-tetra-O-acetyl-α-D-mannopyranose (CAS 65827-58-9)
QUICK LINKS
3-O-Benzyl-1,2,4,6-tetra-O-acetyl-α-D-mannopyranose is a synthetic derivative of mannose that plays a critical role in the field of carbohydrate chemistry, particularly in the synthesis of complex glycostructures. This molecule is equipped with acetyl groups at the 1, 2, 4, and 6 positions and a benzyl group at the 3 position, which serve to protect the hydroxyl groups from undesired side reactions during chemical synthesis. This protection strategy allows for precise and selective modifications, facilitating the creation of specific glycosidic linkages necessary for building oligosaccharides and glycoconjugates. In research, this compound is extensively used as a building block in the assembly of mannose-containing molecules, which are crucial for exploring biological processes such as cell adhesion, microbial recognition, and immune response. The specific arrangement of protective groups enables researchers to systematically expose particular hydroxyl groups by selective deprotection, tailoring the structure to fit the requirements of more complex synthetic targets. Moreover, 3-O-Benzyl-1,2,4,6-tetra-O-acetyl-α-D-mannopyranose is also instrumental in studying enzyme specificity and activity in glycosylation reactions, particularly in the context of developing enzymatic synthesis methods for oligosaccharides. This research contributes significantly to our understanding of the enzymatic mechanisms governing glycosidic bond formation and the role of sugars in biological systems, advancing both fundamental science and applications in biomaterials development.
3-O-Benzyl-1,2,4,6-tetra-O-acetyl-α-D-mannopyranose (CAS 65827-58-9) References
- Glycosylation via locked anomeric configuration: stereospecific synthesis of oligosaccharides containing the beta-D-mannopyranosyl and beta-L-rhamnopyranosyl linkage. | Hodosi, G. and Kovác, P. 1998. Carbohydr Res. 308: 63-75. PMID: 9675357
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
3-O-Benzyl-1,2,4,6-tetra-O-acetyl-α-D-mannopyranose, 200 mg | sc-220877 | 200 mg | $380.00 |
