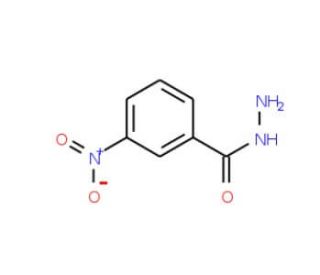
3-Nitrobenzhydrazide (CAS 618-94-0)
QUICK LINKS
3-Nitrobenzhydrazide is a chemical compound that functions as a reagent in organic synthesis. It acts as a precursor in the preparation of various organic compounds, particularly those containing nitrogen functionalities. The mode of action of 3-Nitrobenzhydrazide involves its ability to undergo chemical reactions, such as reduction or substitution, to introduce specific functional groups into target molecules. 3-Nitrobenzhydrazide participates in the formation of new chemical bonds, leading to the synthesis of diverse organic products. Its role in the reaction mechanism involves the transfer of specific chemical moieties, contributing to the transformation of the starting materials into the desired end products. 3-Nitrobenzhydrazide is useful for chemists working on the synthesis of complex organic molecules. Its mode of action at the molecular level involves its reactivity towards other chemical species, leading to the formation of new chemical entities with distinct properties.
3-Nitrobenzhydrazide (CAS 618-94-0) References
- [2-(2,4-Dihydroxyphenyl)-1,3,4-thiadiazole analogues: antifungal activity in vitro against Candida species]. | Matysiak, J. and Malinski, Z. 2007. Bioorg Khim. 33: 640-7. PMID: 18173128
- Predicting the Thermal Stability of Nitroaromatic Compounds Using Chemoinformatic Tools. | Fayet, G., et al. 2011. Mol Inform. 30: 623-34. PMID: 27467162
- Structure-Activity Relationship Analysis of the Thermal Stabilities of Nitroaromatic Compounds Following Different Decomposition Mechanisms. | Li, J., et al. 2013. Mol Inform. 32: 193-202. PMID: 27481280
- Synthesis of 2, 5-disubstituted 1, 3, 4-oxadiazoles from trichloromethylarenes and acylhydrazines. | Poddubnyi, I. S., Belen'skii, L. I., & Krayushkin, M. M. 1994. Chemistry of Heterocyclic Compounds. 30(5): 602-607.
- On the development of QSPR models for regulatory frameworks: The heat of decomposition of nitroaromatics as a test case | Fayet, G., Rotureau, P., & Adamo, C. 2013. Journal of Loss Prevention in the Process Industries. 26(6): 1100-1105.
- A new method for predicting heats of decomposition of nitroaromatics. | Keshavarz, M. H., Ghani, K., & Asgari, A. 2015. Zeitschrift für anorganische und allgemeine Chemie. 641(10): 1818-1823.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
3-Nitrobenzhydrazide, 5 g | sc-276362 | 5 g | $75.00 |
