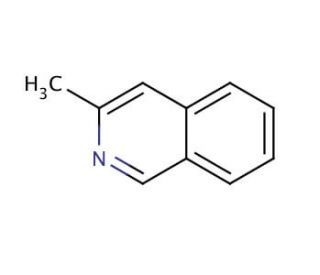
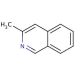
Molecular structure of 3-Methylisoquinoline, CAS Number: 1125-80-0
3-Methylisoquinoline (CAS 1125-80-0)
CAS Number:
1125-80-0
Molecular Weight:
143.19
Molecular Formula:
C10H9N
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
3-Methylisoquinoline is a compound that functions as a building block in organic synthesis. 3-Methylisoquinoline participates in a range of reactions, including Friedel-Crafts acylation, alkylation, and cyclization, contributing to the formation of complex molecular structures. At the molecular level, 3-Methylisoquinoline undergoes transformations that allow for the introduction of specific functional groups, leading to the generation of novel molecules with unique properties. Its role in chemical synthesis contributes to the expansion of chemical libraries and the exploration of new chemical space, potentially leading to the discovery of compounds with distinct biological activities.
3-Methylisoquinoline (CAS 1125-80-0) References
- Microbial production of isoquinoline from indene. | Francis, MM. and Gould, WD. 2003. Can J Microbiol. 49: 699-706. PMID: 14735219
- Molybdenum hydroxylase super family shows circadian activity fluctuation in mice liver: emphasis on aldehyde hydroxylase and xanthine oxidase. | Al-Abbasi, FA. and Al-Sieni, AI. 2010. Pak J Pharm Sci. 23: 359-62. PMID: 20884446
- Some derivatives of 3-methylisoquinoline. | BERGSTROM, FW. and PATERSON, RE. 1945. J Org Chem. 10: 479-82. PMID: 21004585
- New reactions of N-tert-butylimines; formation of N-heterocycles by methyl radical elimination on flash vacuum thermolysis of N-benzylidene- and N-(2-pyridylmethylidene)-tert-butylamines. | Vu, TY., et al. 2013. Chemistry. 19: 14983-8. PMID: 24108670
- Sources of nitrogen heterocyclic PAHs (N-HETs) along a riverine course. | Siemers, AK., et al. 2017. Sci Total Environ. 590-591: 69-79. PMID: 28301813
- Palladium-Catalyzed Isoquinoline Synthesis by Tandem C-H Allylation and Oxidative Cyclization of Benzylamines with Allyl Acetate. | Chen, Y., et al. 2021. Org Lett. 23: 4209-4213. PMID: 33999646
- Analysis of azanaphthalenes and their enzyme oxidation products by high-performance liquid chromatography, infrared spectroscopy and mass spectrometry. | Stubley, C., et al. 1979. J Chromatogr. 177: 313-22. PMID: 528640
- Antagonism of uridine diphosphate with 3-methylisoquinoline compounds. | Haley, TJ. and Christensen, HD. 1965. J Pharm Sci. 54: 1539-40. PMID: 5883238
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
3-Methylisoquinoline, 1 g | sc-223552 | 1 g | $71.00 |
