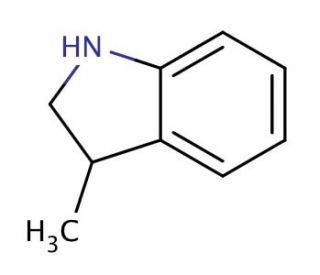
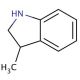
Molecular structure of 3-methylindoline, CAS Number: 4375-15-9
3-methylindoline (CAS 4375-15-9)
CAS Number:
4375-15-9
Molecular Weight:
133.19
Molecular Formula:
C9H11N
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
3-Methylindoline is an organic heterocyclic compound, which are known for their nitrogen-containing rings. This compound has a broad range of applications and is highly versatile, possessing several advantageous properties that make it a preferred choice in laboratory experiments. It is also used as a model compound in the study of biological systems, including enzymes and proteins. Moreover, 3-Methylindoline has been employed to examine the mechanism of action of drugs.
3-methylindoline (CAS 4375-15-9) References
- Highly enantioselective synthesis of chiral 3-substituted indolines by catalytic asymmetric hydrogenation of indoles. | Kuwano, R., et al. 2004. Org Lett. 6: 2213-5. PMID: 15200323
- Synthesis of benzannulated N-heterocycles by a palladium-catalyzed C-C/C-N coupling of bromoalkylamines. | Thansandote, P., et al. 2007. Org Lett. 9: 5255-8. PMID: 18001046
- General approach to the total synthesis of 9-methoxy-substituted indole alkaloids: synthesis of mitragynine, as well as 9-methoxygeissoschizol and 9-methoxy-N(b)-methylgeissoschizol. | Ma, J., et al. 2009. J Org Chem. 74: 264-73. PMID: 19046119
- Redox isomerization via azomethine ylide intermediates: N-alkyl indoles from indolines and aldehydes. | Deb, I., et al. 2011. Org Lett. 13: 812-5. PMID: 21247142
- Intramolecular carbonickelation of alkenes. | Lhermet, R., et al. 2013. Beilstein J Org Chem. 9: 710-6. PMID: 23766783
- Phase-transfer-catalyzed asymmetric synthesis of axially chiral anilides. | Liu, K., et al. 2013. Chem Asian J. 8: 3214-21. PMID: 24273122
- Formal aromaticity transfer for palladium-catalyzed coupling between phenols and pyrrolidines/indolines. | Qiu, Z., et al. 2017. Chem Sci. 8: 6954-6958. PMID: 29147521
- Selective Iron Catalyzed Synthesis of N-Alkylated Indolines and Indoles. | Wu, J., et al. 2022. Chemistry. 28: e202201809. PMID: 35700072
- The pyrolysis of oxindoles at 850°C. II. Oxindoles methylated at N1 and at C3 | RFC Brown, M Butcher. 1973. Australian Journal of Chemistry. 26(2.) 369 - 374.
- Bromo-substituted physostigmine alkaloids from a marine bryozoa Flustra foliacea | . 1979,. J. Am. Chem. Soc. 101, 14,: 4012–4013.
- Routes toward enantiopure 2-substituted indolines: an overview | S Anas, HB Kagan -. October 2009. Tetrahedron: Asymmetry. Volume 20, Issue 19, 6:, Pages 2193-2199.
- Recent advances in the synthetic method and mechanism for the important N-heterocyclic compound of 3-methylindole | . 24 January 2022. Journal of Heterocyclic Chemistry. Volume59, Issue7: Pages 1135-1143.
- Preparation of differentially 1,3-disubstituted indolines by intramolecular carbolithiation | WF Bailey, MR Luderer, MJ Mealy. 7 July 2003, Pages 5303-5305. Tetrahedron Letters. Volume 44, Issue 28,: 7 July 2003, Pages 5303-5305.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
3-methylindoline, 1 g | sc-347182 | 1 g | $435.00 | |||
3-methylindoline, 5 g | sc-347182A | 5 g | $1203.00 |
