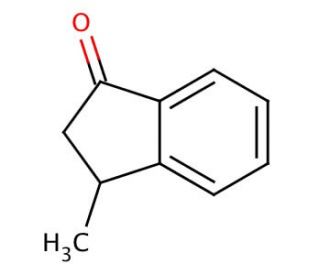
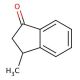
Molecular structure of 3-Methyl-1-indanone, CAS Number: 6072-57-7
3-Methyl-1-indanone (CAS 6072-57-7)
CAS Number:
6072-57-7
Molecular Weight:
146.19
Molecular Formula:
C10H10O
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
3-Methyl-1-indanone is an organic compound classified as a ketone. It presents itself as a colorless liquid with a relatively low boiling point. While the precise mechanism of action of 3-Methyl-1-indanone remains incompletely understood, it is recognized that the compound interacts with specific proteins and enzymes. Additionally, it has the capability to interact with DNA. Furthermore, 3-Methyl-1-indanone has been observed to influence gene expression and impact the activity of certain enzymes.
3-Methyl-1-indanone (CAS 6072-57-7) References
- Efficient kinetic resolution in the asymmetric hydrosilylation of imines of 3-substituted indanones and 4-substituted tetralones. | Yun, J. and Buchwald, SL. 2000. J Org Chem. 65: 767-74. PMID: 10814009
- Theory and use of the pseudophase model in gas-liquid chromatographic enantiomeric separations. | Pino, V., et al. 2006. Anal Chem. 78: 113-9. PMID: 16383317
- Microwave-assisted one-pot synthesis of 1-indanones from arenes and alpha,beta-unsaturated acyl chlorides. | Yin, W., et al. 2006. J Org Chem. 71: 4312-5. PMID: 16709078
- Simulated moving columns technique for enantioselective supercritical fluid chromatography. | Zhang, Y., et al. 2007. Chirality. 19: 683-92. PMID: 17487890
- Unresolved complex mixtures (UCMs) of aromatic hydrocarbons: branched alkyl indanes and branched alkyl tetralins are present in UCMs and accumulated by and toxic to, the mussel Mytilus edulis. | Booth, AM., et al. 2008. Environ Sci Technol. 42: 8122-6. PMID: 19031912
- Synthesis of chiral 3-alkyl-3,4-dihydroisocoumarins by dynamic kinetic resolutions catalyzed by a Baeyer-Villiger monooxygenase. | Rioz-Martínez, A., et al. 2010. J Org Chem. 75: 2073-6. PMID: 20166716
- Stereocontrolled synthesis of substituted chiral piperidines via one-pot asymmetric 6π-azaelectrocyclization: asymmetric syntheses of (-)-dendroprimine, (+)-7-epidendroprimine, (+)-5-epidendroprimine, and (+)-5,7-epidendroprimine. | Kobayashi, T., et al. 2012. J Org Chem. 77: 1812-32. PMID: 22263723
- Photoinduced intramolecular cyclopentanation vs photoprotolytic oxametathesis in polycyclic alkenes outfitted with conformationally constrained aroylmethyl chromophores. | Valiulin, RA., et al. 2013. J Org Chem. 78: 2012-25. PMID: 23106813
- Structure-activity relationship of indoloquinoline analogs anti-MRSA. | Zhao, M., et al. 2015. Bioorg Med Chem Lett. 25: 5551-4. PMID: 26522949
- Current Status on the Functional Characterization of Chemosensory Receptors of Cydia pomonella (Lepidoptera: Tortricidae). | Cattaneo, AM. 2018. Front Behav Neurosci. 12: 189. PMID: 30210318
- Two-Carbon Ring Expansion of 1-Indanones via Insertion of Ethylene into Carbon-Carbon Bonds. | Xia, Y., et al. 2019. J Am Chem Soc. 141: 13038-13042. PMID: 31389237
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
3-Methyl-1-indanone, 1 g | sc-231821 | 1 g | $55.00 |
