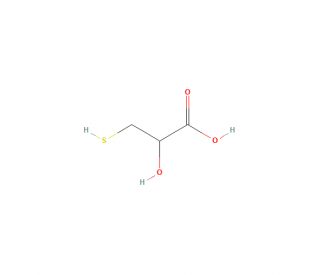
3-Mercaptolactic Acid (CAS 2614-83-7)
QUICK LINKS
3-Mercaptolactic Acid is used in the process of phosphorylation and acetylation of histone H2B. Its thiol group is highly reactive and can participate in a wide range of chemical reactions, including the formation of disulfides, thioesters, and other sulfur-containing compounds. 3-Mercaptolactic acid also finds applications in polymer science, particularly in the synthesis and modification of polymers. The thiol group can undergo radical reactions that facilitate polymerization or cross-linking, leading to materials with unique properties such as enhanced strength, thermal stability, or chemical resistance. This makes 3-mercaptolactic acid useful in the development of coatings, adhesives, and elastomers for various industrial applications.
3-Mercaptolactic Acid (CAS 2614-83-7) References
- Study on the cytochrome P-450- and glutathione-dependent biotransformation of trichloroethylene in humans. | Bloemen, LJ., et al. 2001. Int Arch Occup Environ Health. 74: 102-8. PMID: 11317702
- Determination of aminothiols in body fluids, cells, and tissues by capillary electrophoresis. | Bayle, C., et al. 2004. Electrophoresis. 25: 1457-72. PMID: 15188228
- LC-MS metabolite fingerprinting and MtSK-based screening of an endophyte Bartalinia pondoensis Marinc of Citrus aurantum L. | Zaher, AM., et al. 2014. J Chromatogr B Analyt Technol Biomed Life Sci. 970: 18-23. PMID: 25222744
- Dalmanol biosyntheses require coupling of two separate polyketide gene clusters. | Zhou, ZZ., et al. 2019. Chem Sci. 10: 73-82. PMID: 30746075
- Structure, Synthesis and Inhibition Mechanism of Nucleoside Analogues as HIV-1 Reverse Transcriptase Inhibitors (NRTIs). | Yoshida, Y., et al. 2021. ChemMedChem. 16: 743-766. PMID: 33230979
- NF-κB inhibitory, antimicrobial and antiproliferative potentials of compounds from Hawaiian fungus Aspergillus polyporicola FS910. | Wang, C., et al. 2021. 3 Biotech. 11: 391. PMID: 34458061
- The mechanism of N-acetyl-l-cysteine in improving the secretion of porcine follicle-stimulating hormone in Pichia pastoris. | Luo, G., et al. 2021. Yeast. 38: 601-611. PMID: 34486746
- All Hydroxyl-Thiol-Protected Gold Nanoclusters with Near-Neutral Surface Charge. | Chai, OJH., et al. 2021. J Phys Chem Lett. 12: 9882-9887. PMID: 34609875
- 3-Mercaptopyruvate, 3-mercaptolactate and mercaptoacetate. | Sörbo, B. 1987. Methods Enzymol. 143: 178-82. PMID: 3657534
- Metabolic Changes and Their Associations with Selected Nutrients Intake in the Group of Workers Exposed to Arsenic. | Sijko, M., et al. 2023. Metabolites. 13: PMID: 36676995
- Determination of 3-mercaptolactic acid by amino acid analyzer after aminoethylation. | Pensa, B., et al. 1985. Anal Biochem. 145: 120-3. PMID: 4003755
- Preparation of 3-mercaptolactic acid and S-aminoethylmercaptolactic acid. | Costa, M., et al. 1982. Prep Biochem. 12: 417-27. PMID: 7170304
- Separation of urinary thiols as tributyltinmercaptides and determination using capillary isotachophoresis. | Wroński, M. 1996. J Chromatogr B Biomed Appl. 676: 29-34. PMID: 8852041
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
3-Mercaptolactic Acid, 100 mg | sc-482537 | 100 mg | $408.00 | |||
3-Mercaptolactic Acid, 500 mg | sc-482537D | 500 mg | $1581.00 | |||
3-Mercaptolactic Acid, 1 g | sc-482537A | 1 g | $2703.00 | |||
3-Mercaptolactic Acid, 5 g | sc-482537B | 5 g | $8670.00 | |||
3-Mercaptolactic Acid, 10 g | sc-482537C | 10 g | $12852.00 |
