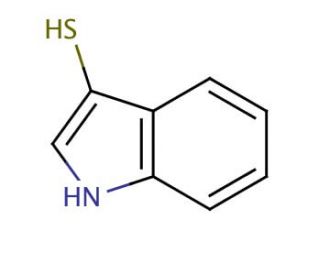
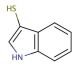
3-Mercaptoindole (CAS 480-94-4)
QUICK LINKS
3-mercaptoindole or 3-thioindole is a widely utilized heterocyclic aromatic organic compound in scientific research, particularly within the field of biochemistry and physiological applications. It serves as a foundational component for synthesizing a diverse range of compounds and finds extensive applications in laboratory applications. This compound holds great significance in scientific research, specifically in biochemistry and physiology, where it has been employed in the synthesis of peptides, proteins, and other biologically active compounds. Additionally it has played a vital role in studying enzyme-catalyzed reactions. Moreover, 1H-Indole-3-thiol has facilitated investigations into cellular signaling pathways and the structure-function relationship of proteins.
3-Mercaptoindole (CAS 480-94-4) References
- Ligand Specificity of Bean Leaf Soluble Auxin-binding Protein. | Wardrop, AJ. and Polya, GM. 1980. Plant Physiol. 66: 112-8. PMID: 16661370
- Potent Activation of Indoleamine 2,3-Dioxygenase by Polysulfides. | Nelp, MT., et al. 2019. J Am Chem Soc. 141: 15288-15300. PMID: 31436417
- Thiocyanation of indole. Some reactions of 3-thiocyanoindole. | Grant, M. S., & Snyder, H. R. 1960. Journal of the American Chemical Society. 82(11): 2742-2744.
- A simple synthesis of amino-containing Bunte salts by the reaction of aminothiols with chlorosulfonic acid | TANAKA, T., NAKAMURA, H., & TAMURA, Z. 1974. Chemical and Pharmaceutical Bulletin. 22(11): 2725-2728.
- Reaction of Skatole with Iodine in the Presence of Thiourea | HINO, T., ENDO, M., & NAKAGAWA, M. 1974. Chemical and Pharmaceutical Bulletin. 22(11): 2728-2731.
- Reaction of 3-mercaptoindole and its ethyl derivatives with acetylene | Skvortsova, G. G., Teterina, L. F., Trzhtsinskaya, B. V., & Voronov, V. K. 1979. Chemistry of Heterocyclic Compounds. 15: 289-291.
- Reactivities of vinyl derivatives of 3-mercaptoindole in reactions with alcohols | Skvortsova, G. G., Trzhtsinskaya, B. V., Chipanina, N. N., Teterina, L. F., & Deriglazov, N. M. 1980. Chemistry of Heterocyclic Compounds. 16: 589-591.
- Derivatives of 3-mercaptoindole-synthesis of a potent vasoconstrictor, 3-(2-imidazolin-2-ylthio) indole (Tinazoline) | Nagarajan, K., Arya, V. P., Parthasarathy, T. N., Shenoy, S. J., Shah, R. K., & Kulkarni, Y. S. 1981. Indian Journal of Chemistry. 20: 672-679.
- Cyclization of 2‐and 3‐indolylthiobenzoic, phenylacetic and nicotinic acids and esters to novel indole‐containing tetracyclic ring systems | Hamel, P., Girard, M., & Tsou, N. N. 1999. Journal of heterocyclic chemistry. 36(3): 643-652.
- Synthesis of N-and S-vinyl derivatives of heteroaromatic compounds using phase-transfer catalysis | Abele, E., Dzenitis, O., Rubina, K., & Lukevics, E. 2002. Chemistry of Heterocyclic Compounds. 38: 682-685.
- Synthesis and Cytotoxicity of 3-(Hetarylthio)-1-propynyl (trimethyl) silanes | Abele, R., Abele, E., Rubina, K., Dzenitis, O., Arsenyan, P., Shestakova, I.,.. & Lukevics, E. 2002. Chemistry of Heterocyclic Compounds. 38: 867-872.
- Ligand-and catalyst-free intramolecular CS bond formation: direct access to indalothiochromen-4-ones | Vijay, T. J., Sandhya, N. C., Pavankumar, C. S., Rangappa, K. S., & Mantelingu, K. 2015. Heterocyclic Communications,. 21(3): 159-163.
- Synthesis and properties of 2, 3-heteroannulated thiochromones-hetero analogs of thioxanthone | Sosnovskikh, V. Y. 2019. Chemistry of Heterocyclic Compounds. 55: 103-125.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
3-Mercaptoindole, 250 mg | sc-216480 | 250 mg | $374.00 |
