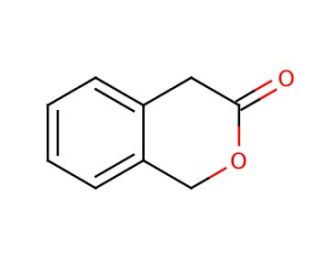
3-Isochromanone (CAS 4385-35-7)
QUICK LINKS
3-Isochromanone (3-ICN) is a naturally occurring heterocyclic aromatic compound that finds application in both scientific research and various industrial endeavors. Its versatility makes it a promising candidate for research and development, owing to its diverse biological effects. Furthermore, it has contributed significantly to studying important biochemical pathways, such as the synthesis of serotonin and the regulation of gene expression. Although the precise mechanism of action for 3-Isochromanone remains elusive, it is believed to involve the inhibition of monoamine oxidase (MAO) enzymes. These enzymes are responsible for breaking down neurotransmitters like serotonin, and by inhibiting them, 3-Isochromanone can lead to elevated serotonin levels. Additionally, this compound may act to interact with other enzymes involved in essential biochemical pathways, like cytochrome P450 enzymes. As research progresses, further insights into the potential of 3-Isochromanone and its applications in both scientific exploration and industry are likely to emerge. Its diverse effects and ability to interact with vital biochemical pathways continue to make it an intriguing subject for future investigations and development.
3-Isochromanone (CAS 4385-35-7) References
- A new isoquinolinone derivative with noble vasorelaxation activity. | Lin, CH., et al. 2003. Pharmacology. 67: 202-10. PMID: 12595751
- Purification and characterization of a novel lactonohydrolase, catalyzing the hydrolysis of aldonate lactones and aromatic lactones, from Fusarium oxysporum. | Shimizu, S., et al. 1992. Eur J Biochem. 209: 383-90. PMID: 1396712
- Analysis of the vibrational spectra of new OH-containing E-4-arylmethylene-3-isochromanones and 3-arylcoumarins. | Keresztury, G., et al. 2006. J Biochem Biophys Methods. 69: 163-77. PMID: 16624418
- Reactions of 3-isochromanone with aromatic aldehydes--microwave assisted condensations performed on solid basic inorganic supports. | Vass, A., et al. 2006. J Biochem Biophys Methods. 69: 179-87. PMID: 16678272
- Activation and functionalization of benzylic derivatives by palladium catalysts. | Liégault, B., et al. 2008. Chem Soc Rev. 37: 290-9. PMID: 18197345
- Luminescence enhancement of pyrene/dispersant nanoarrays driven by the nanoscale spatial effect on mixing. | Lo, KH., et al. 2013. Langmuir. 29: 1627-33. PMID: 23293950
- Pd(II)-catalyzed decarboxylative acylation of phenylacetamides with α-oxocarboxylic acids via C-H bond activation. | Park, J., et al. 2013. Chem Commun (Camb). 49: 1654-6. PMID: 23334232
- Catalytic Asymmetric Conjugate Addition of Carboxylic Acids via Oxa-Michael Reaction of Peroxy Hemiacetals followed by Kornblum DeLaMare Fragmentation. | Parhi, B., et al. 2016. Org Lett. 18: 5220-5223. PMID: 27690466
- Enantioselective Synthesis of α-Allyl-α-aryldihydrocoumarins and 3-Isochromanones via Pd-Catalyzed Decarboxylative Asymmetric Allylic Alkylation. | Akula, R. and Guiry, PJ. 2016. Org Lett. 18: 5472-5475. PMID: 27780358
- Rhodium(III)-Catalyzed Redox-Neutral Weak O-Coordinating Vinylation and Allylation of Arylacetamides with Allylic Acetates. | Jambu, S. and Jeganmohan, M. 2019. Org Lett. 21: 5655-5659. PMID: 31283249
- Novel Cell Wall Antifungals Reveal a Special Synergistic Activity in pbr1 Mutants Resistant to the Glucan Synthesis Antifungals Papulacandins and Echinocandins. | Berzaghi, R., et al. 2019. Front Microbiol. 10: 1692. PMID: 31428061
- Radical ring-opening polymerization of sustainably-derived thionoisochromanone. | Prebihalo, EA., et al. 2023. Chem Sci. 14: 5689-5698. PMID: 37265728
- New metabolites in the degradation of fluorene by Arthrobacter sp. strain F101. | Casellas, M., et al. 1997. Appl Environ Microbiol. 63: 819-26. PMID: 9055403
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
3-Isochromanone, 1 g | sc-231776 | 1 g | $31.00 |
