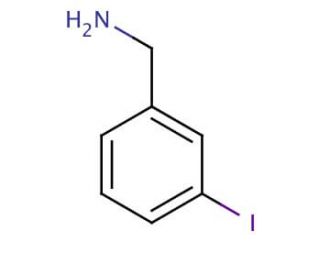
Molecular structure of 3-Iodobenzylamine, CAS Number: 696-40-2
3-Iodobenzylamine (CAS 696-40-2)
CAS Number:
696-40-2
Molecular Weight:
233.05
Molecular Formula:
C7H8IN
Supplemental Information:
This is classified as a Dangerous Good for transport and may be subject to additional shipping charges.
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
3-Iodobenzylamine functions as a versatile building block in organic synthesis. It acts as a nucleophilic reagent in various reactions, including nucleophilic aromatic substitution and palladium-catalyzed cross-coupling reactions. 3-Iodobenzylamine′s mechanism of action involves its ability to undergo substitution reactions at the iodine atom, leading to the formation of new carbon-nitrogen bonds. This allows for the introduction of the 3-Iodobenzylamine moiety into complex organic molecules, enabling the synthesis of diverse chemical structures. Its reactivity and selectivity make it useful in the construction of complex molecular architectures and the development of novel chemical entities.
3-Iodobenzylamine (CAS 696-40-2) References
- Modulation of adenosine receptor affinity and intrinsic efficacy in adenine nucleosides substituted at the 2-position. | Ohno, M., et al. 2004. Bioorg Med Chem. 12: 2995-3007. PMID: 15142558
- Exploring human adenosine A3 receptor complementarity and activity for adenosine analogues modified in the ribose and purine moiety. | Van Rompaey, P., et al. 2005. Bioorg Med Chem. 13: 973-83. PMID: 15670905
- (N)-methanocarba 2,N6-disubstituted adenine nucleosides as highly potent and selective A3 adenosine receptor agonists. | Tchilibon, S., et al. 2005. J Med Chem. 48: 1745-58. PMID: 15771421
- Purine derivatives as ligands for A3 adenosine receptors. | Joshi, BV. and Jacobson, KA. 2005. Curr Top Med Chem. 5: 1275-95. PMID: 16305531
- Orthogonal activation of the reengineered A3 adenosine receptor (neoceptor) using tailored nucleoside agonists. | Gao, ZG., et al. 2006. J Med Chem. 49: 2689-702. PMID: 16640329
- Synthesis of 3'-acetamidoadenosine derivatives as potential A3 adenosine receptor agonists. | Chun, MW., et al. 2008. Nucleosides Nucleotides Nucleic Acids. 27: 408-20. PMID: 18404574
- Regio- and stereoselective synthesis of truncated 3'-aminocarbanucleosides and their binding affinity at the A3 adenosine receptor. | Choi, MJ., et al. 2011. Org Biomol Chem. 9: 6955-62. PMID: 21860878
- Synthesis and Binding Affinity of Homologated Adenosine Analogues as A3 Adenosine Receptor Ligands. | Lee, HW., et al. 2011. Bull Korean Chem Soc. 32: 1620-1624. PMID: 26120223
- N6-Substituted 5'-N-Methylcarbamoyl-4'-selenoadenosines as Potent and Selective A3 Adenosine Receptor Agonists with Unusual Sugar Puckering and Nucleobase Orientation. | Yu, J., et al. 2017. J Med Chem. 60: 3422-3437. PMID: 28380296
- Polypharmacology of N6-(3-Iodobenzyl)adenosine-5'-N-methyluronamide (IB-MECA) and Related A3 Adenosine Receptor Ligands: Peroxisome Proliferator Activated Receptor (PPAR) γ Partial Agonist and PPARδ Antagonist Activity Suggests Their Antidiabetic Potential. | Yu, J., et al. 2017. J Med Chem. 60: 7459-7475. PMID: 28799755
- Recent Advances in the Synthesis of Peptoid Macrocycles. | Webster, AM. and Cobb, SL. 2018. Chemistry. 24: 7560-7573. PMID: 29356125
- Epidermal Growth Factor Receptor-Targeted Multifunctional Photosensitizers for Bladder Cancer Imaging and Photodynamic Therapy. | Cheruku, RR., et al. 2019. J Med Chem. 62: 2598-2617. PMID: 30776232
- Subtle Chemical Changes Cross the Boundary between Agonist and Antagonist: New A3 Adenosine Receptor Homology Models and Structural Network Analysis Can Predict This Boundary. | Lee, Y., et al. 2021. J Med Chem. 64: 12525-12536. PMID: 34435786
- Sulfur-Phenolate Exchange as a Mild, Fast, and High-Yielding Method toward the Synthesis of Sulfonamides. | van den Boom, AFJ. and Zuilhof, H. 2023. Org Lett. 25: 788-793. PMID: 36720015
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
3-Iodobenzylamine, 1 g | sc-226102 | 1 g | $95.00 |
