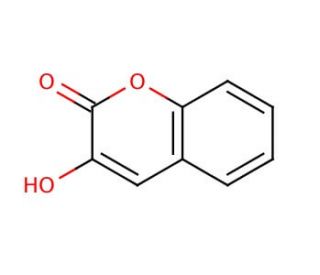
3-Hydroxycoumarin (CAS 939-19-5)
QUICK LINKS
3-Hydroxycoumarin is a compound that functions as a fluorescent probe in biochemical and cellular assays. It is used to detect and measure the presence of metal ions, particularly iron, in biological samples. The compound chelates with metal ions, leading to a change in its fluorescence properties, allowing for the quantification of metal ion concentrations. 3-Hydroxycoumarin also acts as a photosensitizer, where it generates reactive oxygen species upon exposure to light, leading to cell death. 3-Hydroxycoumarin′s mechanism of action involves its ability to bind to metal ions and undergo photochemical reactions, which may be useful for studying metal ion homeostasis and for inducing cell death in experimental applications.
3-Hydroxycoumarin (CAS 939-19-5) References
- o-hydroxyphenylacetaldehyde is a hepatotoxic metabolite of coumarin. | Born, SL., et al. 2000. Drug Metab Dispos. 28: 218-23. PMID: 10640521
- Cloning and characterization of a glucosyltransferase that reacts on 7-hydroxyl group of flavonol and 3-hydroxyl group of coumarin from tobacco cells. | Taguchi, G., et al. 2003. Arch Biochem Biophys. 420: 95-102. PMID: 14622979
- Metabolism of coumarin by rat, gerbil and human liver microsomes. | Fentem, JH. and Fry, JR. 1992. Xenobiotica. 22: 357-67. PMID: 1496825
- 3-Hydroxycoumarin as a new matrix for matrix-assisted laser desorption/ionization time-of-flight mass spectrometry of DNA. | Zhang, Z., et al. 2006. J Am Soc Mass Spectrom. 17: 1665-8. PMID: 16908181
- Effect of inducers of cytochrome P-450 on the metabolism of [3-14C]coumarin by rat hepatic microsomes. | Peters, MM., et al. 1991. Xenobiotica. 21: 499-514. PMID: 1897250
- Photoprotective effect of coumarin and 3-hydroxycoumarin in sea urchin gametes and embryonic cells. | de Araujo Leite, JC., et al. 2015. J Photochem Photobiol B. 146: 44-51. PMID: 25795999
- Structure-Activity Relationship Study of Hydroxycoumarins and Mushroom Tyrosinase. | Asthana, S., et al. 2015. J Agric Food Chem. 63: 7236-44. PMID: 26263396
- Origin of Remarkably Different Acidity of Hydroxycoumarins-Joint Experimental and Theoretical Studies. | Nowak, PM., et al. 2017. J Phys Chem B. 121: 4554-4561. PMID: 28406631
- A novel class of human 15-LOX-1 inhibitors based on 3-hydroxycoumarin. | Alavi, SJ., et al. 2018. Chem Biol Drug Des. 91: 1125-1132. PMID: 29388345
- 3-hydroxycoumarin loaded vesicles for recombinant human tyrosinase inhibition in topical applications. | Schlich, M., et al. 2018. Colloids Surf B Biointerfaces. 171: 675-681. PMID: 30107341
- Identification and Characterization of Two New 1- O-Acyl-glucose-ester Forming Glucosyltransferases from Erigeron breviscapus. | Yang, Y., et al. 2019. J Agric Food Chem. 67: 2848-2855. PMID: 30821967
- Achillea fragrantissima (Forssk.) Sch.Bip. methanolic extract exerts potent antimicrobial activity and causes cancer cell death via induction of caspase-dependent apoptosis and S-phase arrest. | Break, MKB., et al. 2022. Nat Prod Res. 36: 4645-4650. PMID: 34847782
- Coumarin derivatives as inhibitors of d-amino acid oxidase and monoamine oxidase. | Bester, E., et al. 2022. Bioorg Chem. 123: 105791. PMID: 35413582
- The complex photochemistry of coumarin-3-carboxylic acid in acetonitrile and methanol. | Yang, Q., et al. 2022. Photochem Photobiol Sci. 21: 1481-1495. PMID: 35578152
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
3-Hydroxycoumarin, 1 g | sc-231751 | 1 g | $22.00 |
