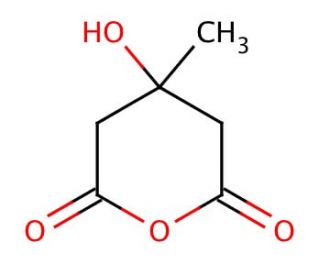
3-Hydroxy-3-methylglutaric Anhydride (CAS 34695-32-4)
See product citations (1)
QUICK LINKS
3-Hydroxy-3-methylglutaric anhydride, often abbreviated as HMG-CoA, plays a pivotal role in the mevalonate pathway, a metabolic route responsible for the synthesis of several essential biomolecules, including cholesterol, steroid hormones, and various isoprenoids. Within this pathway, 3-Hydroxy-3-methylglutaric Anhydride acts as a key intermediate; it is synthesized from acetyl-CoA and acetoacetyl-CoA through an enzyme-catalyzed reaction. The enzyme 3-Hydroxy-3-methylglutaric Anhydride synthase catalyzes this reaction, marking a critical step in the pathway. Subsequently, 3-Hydroxy-3-methylglutaric Anhydride reductase, another highly regulated enzyme, catalyzes the reduction of 3-Hydroxy-3-methylglutaric Anhydride to mevalonic acid, a reaction that is considered the rate-limiting step in the synthesis of cholesterol. The regulation of this step is of paramount importance in controlling the overall pathway flux. In research applications, 3-Hydroxy-3-methylglutaric Anhydride and its interactions are extensively studied to understand cellular lipid metabolism, the regulation of cholesterol biosynthesis, and the mechanisms underlying the synthesis of isoprenoids. These studies are vital for unraveling the complex network of biochemical pathways that sustain cellular function and for developing strategies to manipulate these pathways for various biotechnological applications.
3-Hydroxy-3-methylglutaric Anhydride (CAS 34695-32-4) References
- Substrate and inhibitor specificity of 3-hydroxy-3-methylglutaryl-CoA reductase determined with substrate-analogue CoA-thioesters and CoA-thioethers. | Hupperich, M., et al. 1991. Biol Chem Hoppe Seyler. 372: 857-63. PMID: 1772599
- A Class of Reactive Acyl-CoA Species Reveals the Non-enzymatic Origins of Protein Acylation. | Wagner, GR., et al. 2017. Cell Metab. 25: 823-837.e8. PMID: 28380375
- Crystal structures of the mitochondrial deacylase Sirtuin 4 reveal isoform-specific acyl recognition and regulation features. | Pannek, M., et al. 2017. Nat Commun. 8: 1513. PMID: 29138502
- Isolation and purification of 3-hydroxy-3-methylglutaryl-coenzyme A by ion-exchange chromatography. | Williamson, IP. and Rodwell, VW. 1981. J Lipid Res. 22: 184-7. PMID: 6163836
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
3-Hydroxy-3-methylglutaric Anhydride, 250 mg | sc-206648 | 250 mg | $714.00 |
