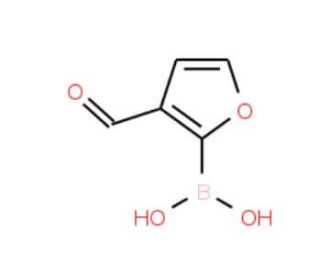
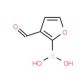
3-Formylfuran-2-boronic acid (CAS 27339-38-4)
QUICK LINKS
3-Formylfuran-2-boronic acid (3F2BA) holds significant importance as an organoboron compound. Its versatility as a reagent enables the synthesis of heterocyclic compounds and various other organic molecules. 3-Formylfuran-2-boronic acid has been widely employed in diverse chemical transformations, including Suzuki-Miyaura cross-coupling reactions, palladium-catalyzed reactions, and direct C-H functionalization reactions. The applications of 3-Formylfuran-2-boronic acid span across scientific research endeavors. Its utilization encompasses the synthesis of heterocyclic compounds and polymers. Additionally, it plays a role in the synthesis of nanomaterials such as carbon nanotubes and graphene. Furthermore, 3-Formylfuran-2-boronic acid contributes to the synthesis of metal-organic frameworks (MOFs), which find wide-ranging applications in catalysis, sensing, and drug delivery. As a versatile reagent, 3-Formylfuran-2-boronic acid partakes in a multitude of chemical transformations. In one such reaction, the direct reaction of furan-2-boronic acid with formic acid, formic acid assumes the role of a nucleophile while the boron atom of furan-2-boronic acid acts as an electrophile. The reaction progresses through several steps, involving a nucleophilic attack of formic acid on the boron atom, subsequent proton transfer, and the formation of a boron-carbon bond.
3-Formylfuran-2-boronic acid (CAS 27339-38-4) References
- Suzuki reaction of vinyl triflates from six- and seven-membered N-alkoxycarbonyl lactams with boronic acids and esters. | Occhiato, EG., et al. 2001. J Org Chem. 66: 2459-65. PMID: 11281788
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
3-Formylfuran-2-boronic acid, 1 g | sc-261003 | 1 g | $160.00 | |||
3-Formylfuran-2-boronic acid, 5 g | sc-261003A | 5 g | $681.00 |
