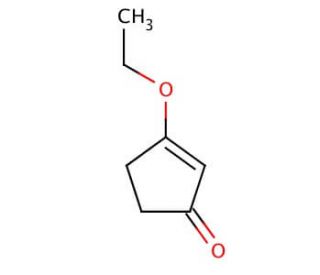
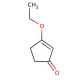
3-Ethoxy-2-cyclopentenone (CAS 22627-70-9)
QUICK LINKS
3-Ethoxy-2-cyclopentenone is a specialized organic compound, notable for its unique structure that includes an ethoxy group attached to a cyclopentenone ring. This configuration endows it with significant chemical reactivity, particularly useful in synthetic organic chemistry. The compound features an α,β-unsaturated carbonyl group, which makes it an excellent electrophile for nucleophilic addition reactions, as well as a potent dienophile suitable for Diels-Alder reactions—key processes in constructing complex organic structures. In research settings, 3-ethoxy-2-cyclopentenone has been utilized to explore innovative synthetic routes and to study reaction mechanisms that can lead to the development of novel compounds with potential applications in materials science and organic synthesis. Its ability to participate in various organic reactions helps explain the dynamics of chemical interactions and reaction pathways, significantly contributing to the field of synthetic chemistry through the development of new methodologies and the improvement of reaction efficiency.
3-Ethoxy-2-cyclopentenone (CAS 22627-70-9) References
- Sulfoxide-Mediated Asymmetric Synthesis of Glycosidase Inhibitor Precursors. | Bueno, AB., et al. 1997. J Org Chem. 62: 2139-2143. PMID: 11671519
- Stereocontrolled synthesis of carbocyclic compounds with a quaternary carbon atom based on SN2' alkylation of γ,δ-epoxy-α,β-unsaturated ketones. | Yoshimura, F., et al. 2012. Org Biomol Chem. 10: 5431-42. PMID: 22706976
- Ring Opening of Bicyclo[3.1.0]hexan-2-ones: A Versatile Synthetic Platform for the Construction of Substituted Benzoates. | Feierfeil, J., et al. 2015. Angew Chem Int Ed Engl. 54: 11835-8. PMID: 26255867
- A 16-step synthesis of the isoryanodane diterpene (+)-perseanol. | Han, A., et al. 2019. Nature. 573: 563-567. PMID: 31554978
- Synthesis of Complex Diterpenes: Strategies Guided by Oxidation Pattern Analysis. | Dibrell, SE., et al. 2021. Acc Chem Res. 54: 1360-1373. PMID: 33621061
- Prostaglandin inhibition of innervated antral motility in dogs. | Chawla, RC. and Eisenberg, MM. 1969. Proc Soc Exp Biol Med. 132: 1081-6. PMID: 5360985
- The Pepsin-Catalyzed Hydrolysis of N-Acetyl-L-phenylalanyl-L-3, 5-dibromotyrosine at pH 2 | Zeffren, E., & Kaiser, E. T. 1966. Journal of the American Chemical Society. 88(13): 3129-3131.
- Palladium-catalyzed intermolecular vinylic arylation of cycloalkenes. Applications to the synthesis of quinolone antibacterials | Laborde, E., Lesheski, L. E., & Kiely, J. S. 1990. Tetrahedron letters. 31(13): 1837-1840.
- Pressure‐Induced Diastereoselectivity in Photochemical [2+ 2] Cycloaddition Reactions | Buback, M., Bünger, J., & Tietze, L. F. 1992. Chemische Berichte. 125(11): 2577-2582.
- Manganese (III) acetate based tandem oxidation of various cyclic β-alkoxy α, β-unsaturated ketones | Tanyeli, C., & Sezen, B. 2000. Tetrahedron Letters. 41(41): 7973-7976.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
3-Ethoxy-2-cyclopentenone, 5 g | sc-238537 | 5 g | $313.00 |
