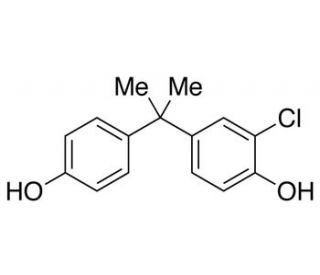
3-Chlorobisphenol A (CAS 74192-35-1)
See product citations (2)
QUICK LINKS
3-Chlorobisphenol A functions as a potential endocrine disruptor in experimental applications. It is known to interact with hormone receptors, particularly estrogen receptors, affecting their normal function. 3-Chlorobisphenol A may bind to estrogen receptors, leading to the disruption of normal hormonal signaling pathways. 3-Chlorobisphenol A has been observed to interfere with gene expression related to hormone regulation, potentially impacting cellular processes. Its mechanism of action involves altering the activity of estrogen receptors, which can have implications for various biological processes. In experimental applications, 3-Chlorobisphenol A is studied for its ability to interfere with endocrine function and its potential impact on cellular and molecular processes.
3-Chlorobisphenol A (CAS 74192-35-1) References
- Characteristics of a thyroid hormone responsive reporter gene transduced into a Xenopus laevis cell line using lentivirus vector. | Sugiyama, S., et al. 2005. Gen Comp Endocrinol. 144: 270-9. PMID: 16102758
- Non-genomic effects of endocrine disrupters: inhibition of estrogen sulfotransferase by phenols and chlorinated phenols. | Harris, RM., et al. 2005. Mol Cell Endocrinol. 244: 72-4. PMID: 16266779
- Change of estrogenic activity and release of chloride ion in chlorinated bisphenol a after exposure to ultraviolet B. | Mutou, Y., et al. 2006. Biol Pharm Bull. 29: 2116-9. PMID: 17015961
- Induction of apoptosis by UV-irradiated chlorinated bisphenol A in Jurkat cells. | Mutou, Y., et al. 2008. Toxicol In Vitro. 22: 864-72. PMID: 18280695
- UVB-exposed chlorinated bisphenol A generates phosphorylated histone H2AX in human skin cells. | Ibuki, Y., et al. 2008. Chem Res Toxicol. 21: 1770-6. PMID: 18729329
- Assessment of thyroid hormone activity of halogenated bisphenol A using a yeast two-hybrid assay. | Terasaki, M., et al. 2011. Chemosphere. 84: 1527-30. PMID: 21550628
- Chemical change of chlorinated bisphenol A by ultraviolet irradiation and cytotoxicity of their products on Jurkat cells. | Mutou, Y., et al. 2006. Environ Toxicol Pharmacol. 21: 283-9. PMID: 21783670
- New insight into photo-bromination processes in saline surface waters: the case of salicylic acid. | Tamtam, F. and Chiron, S. 2012. Sci Total Environ. 435-436: 345-50. PMID: 22863810
- Human Exposures to Bisphenol A, Bisphenol F and Chlorinated Bisphenol A Derivatives and Thyroid Function. | Andrianou, XD., et al. 2016. PLoS One. 11: e0155237. PMID: 27783680
- Agonistic effects of diverse xenobiotics on the constitutive androstane receptor as detected in a recombinant yeast-cell assay. | Kamata, R., et al. 2018. Toxicol In Vitro. 46: 335-349. PMID: 28927721
- Formation and enhanced photodegradation of chlorinated derivatives of bisphenol A in wastewater treatment plant effluent. | Wan, D., et al. 2020. Water Res. 184: 116002. PMID: 32682078
- Broadening the lens on bisphenols in coastal waters: Occurrence, partitioning, and input fluxes of multiple novel bisphenol S derivatives along with BPA and BPA analogues in the Pearl River Delta, China. | Liang, X., et al. 2023. Environ Pollut. 322: 121194. PMID: 36738885
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
3-Chlorobisphenol A, 5 mg | sc-206638 | 5 mg | $347.00 |
