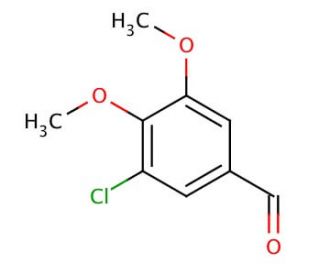
3-Chloro-4,5-dimethoxybenzaldehyde (CAS 18268-68-3)
QUICK LINKS
3-Chloro-4,5-dimethoxybenzaldehyde (3C4DMB) stands as an aromatic aldehyde with wide-ranging applications in organic synthesis and biochemistry. It serves as a versatile building block for synthesizing various molecules, including agrochemicals, and acts as a starting material for heterocyclic compound synthesis. The highly reactive nature of 3-Chloro-4,5-dimethoxybenzaldehyde allows its utilization as a reagent in organic synthesis and biochemistry. Within organic synthesis, it enables the formation of diverse compounds, including aryl aldehydes, alkynes, and heterocyclic compounds. Moreover, in biochemistry, it plays a pivotal role in the synthesis of diverse enzymes, including cytochrome P450 and β-lactamases. Understanding the mechanism of action of 3-Chloro-4,5-dimethoxybenzaldehyde is contingent upon its specific application. In the context of organic synthesis, it acts as a reagent, facilitating the formation of an array of compounds. In biochemistry, it serves as a precursor for synthesizing enzymes, such as cytochrome P450 and β-lactamases.
3-Chloro-4,5-dimethoxybenzaldehyde (CAS 18268-68-3) References
- Synthesis and cytotoxic properties of novel (E)-3-benzylidene-7-methoxychroman-4-one derivatives. | Noushini, S., et al. 2013. Daru. 21: 31. PMID: 23587260
- Multimodal 4-arylchromene derivatives with microtubule-destabilizing, anti-angiogenic, and MYB-inhibitory activities. | Köhler, LHF., et al. 2023. Cancer Drug Resist. 6: 59-77. PMID: 37065868
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
3-Chloro-4,5-dimethoxybenzaldehyde, 1 g | sc-226020 | 1 g | $136.00 |
