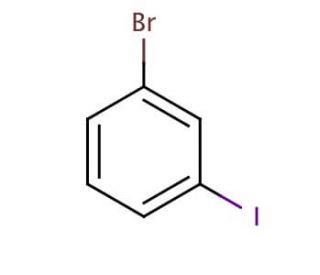
Molecular structure of 3-Bromoiodobenzene, CAS Number: 591-18-4
3-Bromoiodobenzene (CAS 591-18-4)
CAS Number:
591-18-4
Molecular Weight:
282.90
Molecular Formula:
BrC6H4I
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
3-Bromoiodobenzene serves as a versatile building block in organic synthesis. It functions as a precursor for the manufacture of agrochemicals and various materials. 3-Bromoiodobenzene participates in substitution reactions, in palladium-catalyzed cross-coupling reactions, where it undergoes halogen-metal exchange to form carbon-carbon or carbon-heteroatom bonds. Through these reactions, 3-Bromoiodobenzene facilitates the introduction of specific functional groups into organic molecules, enabling the creation of diverse chemical structures. Its mechanism of action involves the activation of the carbon-iodine bond, allowing for the formation of new chemical bonds with high selectivity and efficiency.
3-Bromoiodobenzene (CAS 591-18-4) References
- Auxiliary-assisted palladium-catalyzed arylation and alkylation of sp2 and sp3 carbon-hydrogen bonds. | Shabashov, D. and Daugulis, O. 2010. J Am Chem Soc. 132: 3965-72. PMID: 20175511
- Triplet States and electronic relaxation in photoexcited graphene quantum dots. | Mueller, ML., et al. 2010. Nano Lett. 10: 2679-82. PMID: 20575573
- Asymmetric Au-catalyzed cycloisomerization of 1,6-enynes: An entry to bicyclo[4.1.0]heptene. | Pradal, A., et al. 2011. Beilstein J Org Chem. 7: 1021-9. PMID: 21915203
- Rhodanine and thiohydantoin derivatives for detecting tau pathology in Alzheimer's brains. | Ono, M., et al. 2011. ACS Chem Neurosci. 2: 269-75. PMID: 22778869
- Substrate-based fragment identification for the development of selective, nonpeptidic inhibitors of striatal-enriched protein tyrosine phosphatase. | Baguley, TD., et al. 2013. J Med Chem. 56: 7636-50. PMID: 24083656
- Structure-Activity Study of Bioisosteric Trifluoromethyl and Pentafluorosulfanyl Indole Inhibitors of the AAA ATPase p97. | Alverez, C., et al. 2015. ACS Med Chem Lett. 6: 1225-30. PMID: 26713109
- H-Bond Self-Assembly: Folding versus Duplex Formation. | Núñez-Villanueva, D., et al. 2017. J Am Chem Soc. 139: 6654-6662. PMID: 28470070
- General Synthetic Method for Si-Fluoresceins and Si-Rhodamines. | Grimm, JB., et al. 2017. ACS Cent Sci. 3: 975-985. PMID: 28979939
- X-ray Characterization and Structure-Based Optimization of Striatal-Enriched Protein Tyrosine Phosphatase Inhibitors. | Witten, MR., et al. 2017. J Med Chem. 60: 9299-9319. PMID: 29116812
- New Developments on the Hirao Reactions, Especially from 'Green' Point of View. | Henyecz, R. and Keglevich, G. 2019. Curr Org Synth. 16: 523-545. PMID: 31984929
- Rational Design of Bioavailable Photosensitizers for Manipulation and Imaging of Biological Systems. | Binns, TC., et al. 2020. Cell Chem Biol. 27: 1063-1072.e7. PMID: 32698018
- Gold(I)-Catalyzed Domino Reaction for Furopyrans Synthesis. | Ruch, M., et al. 2020. Molecules. 25: PMID: 33121149
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
3-Bromoiodobenzene, 5 g | sc-231550 | 5 g | $20.00 |
