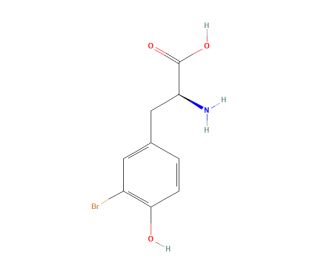
Molecular structure of 3-Bromo-L-tyrosine
3-Bromo-L-tyrosine (CAS 38739-13-8)
CAS Number:
38739-13-8
Molecular Weight:
260.08
Molecular Formula:
C9H10BrNO3
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
3-Bromo-L-tyrosine is a derivative of tyrosine, an essential amino acid, and has been widely used in the synthesis of various compounds, including peptides, proteins, and pharmaceuticals. It can interact with enzymes, DNA, and proteins, thus affecting their activity, structure, and function. 3-Bromo-L-tyrosine has been utilized to study the effects of bromination on the metabolism of drugs, enzyme activity, and the structure and function of proteins and DNA.
3-Bromo-L-tyrosine (CAS 38739-13-8) References
- Changing the amino acid specificity of yeast tyrosyl-tRNA synthetase by genetic engineering. | Ohno, S., et al. 2001. J Biochem. 130: 417-23. PMID: 11530018
- Metabolism of l-Tyrosine to 4-Hydroxybenzaldehyde and 3-Bromo-4-Hydroxybenzaldehyde by Chloroplast-containing Fractions of Odonthalia floccosa (Esp.) Falk. | Manley, SL. and Chapman, DJ. 1979. Plant Physiol. 64: 1032-8. PMID: 16661087
- Aminoacrylate intermediates in the reaction of Citrobacter freundii tyrosine phenol-lyase. | Phillips, RS., et al. 2006. Biochemistry. 45: 9575-83. PMID: 16878992
- Chemistry of unprotected amino acids in aqueous solution: direct bromination of aromatic amino acids with bromoisocyanuric acid sodium salt under strong acidic condition. | Yokoyama, Y., et al. 2006. Chem Pharm Bull (Tokyo). 54: 1715-9. PMID: 17139108
- Functional replacement of the endogenous tyrosyl-tRNA synthetase-tRNATyr pair by the archaeal tyrosine pair in Escherichia coli for genetic code expansion. | Iraha, F., et al. 2010. Nucleic Acids Res. 38: 3682-91. PMID: 20159998
- Preparation of 3-bromo-L-tyrosine and 3,5-dibromo-L-tyrosine. | Phillips, RS., et al. 2013. Amino Acids. 44: 529-32. PMID: 22836678
- Use of Bromine and Bromo-Organic Compounds in Organic Synthesis. | Saikia, I., et al. 2016. Chem Rev. 116: 6837-7042. PMID: 27199233
- Functional analysis of iodotyrosine deiodinase from drosophila melanogaster. | Phatarphekar, A. and Rokita, SE. 2016. Protein Sci. 25: 2187-2195. PMID: 27643701
- Engineering an Automaturing Transglutaminase with Enhanced Thermostability by Genetic Code Expansion with Two Codon Reassignments. | Ohtake, K., et al. 2018. ACS Synth Biol. 7: 2170-2176. PMID: 30063837
- In vivo biosynthesis of tyrosine analogs and their concurrent incorporation into a residue-specific manner for enzyme engineering. | Won, Y., et al. 2019. Chem Commun (Camb). 55: 15133-15136. PMID: 31789331
- Simultaneous LC-MS/MS-Based Quantification of Free 3-Nitro-l-tyrosine, 3-Chloro-l-tyrosine, and 3-Bromo-l-tyrosine in Plasma of Colorectal Cancer Patients during Early Postoperative Period. | Fleszar, MG., et al. 2020. Molecules. 25: PMID: 33167555
- Recent advancements in enzyme engineering via site-specific incorporation of unnatural amino acids. | Zhu, HQ., et al. 2021. World J Microbiol Biotechnol. 37: 213. PMID: 34741210
- A demonstration of two-component behavior of the circular dichroic spectra of poly (L-tyrosine, 3-bromo-L-tyrosine) in trimethylphosphate. | Applequist, J. and Lunde, BK. 1974. Biopolymers. 13: 1341-6. PMID: 4411623
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
3-Bromo-L-tyrosine, 1 g | sc-504311 | 1 g | $296.00 |
