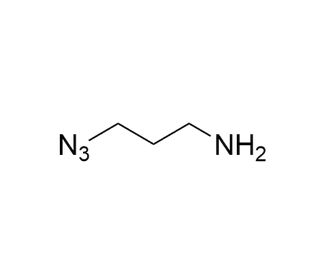
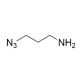
3-Azido-1-propanamine (CAS 88192-19-2)
QUICK LINKS
3-Azido-1-propanamine is a chemical compound utilized in the field of organic synthesis and materials science. Its azido group makes it a reagent for click chemistry reactions, particularly the Huisgen cycloaddition, which allows for the creation of 1,2,3-triazoles. This reaction is of high interest in constructing complex molecular architectures with precision. 3-Azido-1-propanamine is involved in polymer where it can be used to introduce azide functional groups into polymers, which subsequently can be utilized for further functionalization or to alter the properties of the material. It plays a role in the synthesis of heterocyclic compounds, which are important in the development of new materials with potential applications in electronics and optics.
3-Azido-1-propanamine (CAS 88192-19-2) References
- Dual display of proteins on the yeast cell surface simplifies quantification of binding interactions and enzymatic bioconjugation reactions. | Lim, S., et al. 2017. Biotechnol J. 12: PMID: 28299901
- Genetically Encoded Quinone Methides Enabling Rapid, Site-Specific, and Photocontrolled Protein Modification with Amine Reagents. | Liu, J., et al. 2020. J Am Chem Soc. 142: 17057-17068. PMID: 32915556
- Picomolar SARS-CoV-2 Neutralization Using Multi-Arm PEG Nanobody Constructs. | Moliner-Morro, A., et al. 2020. Biomolecules. 10: PMID: 33322557
- Cascaded amplification of intracellular oxidative stress and reversion of multidrug resistance by nitric oxide prodrug based-supramolecular hydrogel for synergistic cancer chemotherapy. | Zhang, J., et al. 2021. Bioact Mater. 6: 3300-3313. PMID: 33778206
- Size- and Surface- Dual Engineered Small Polyplexes for Efficiently Targeting Delivery of siRNA. | Liu, S., et al. 2021. Molecules. 26: PMID: 34072265
- A structure-specific small molecule inhibits a miRNA-200 family member precursor and reverses a type 2 diabetes phenotype. | Haniff, HS., et al. 2022. Cell Chem Biol. 29: 300-311.e10. PMID: 34320373
- High-quantum yield alloy-typed core/shell CdSeZnS/ZnS quantum dots for bio-applications. | Kim, J., et al. 2022. J Nanobiotechnology. 20: 22. PMID: 34991619
- A bispecific monomeric nanobody induces spike trimer dimers and neutralizes SARS-CoV-2 in vivo. | Hanke, L., et al. 2022. Nat Commun. 13: 155. PMID: 35013189
- Desymmetrization via Activated Esters Enables Rapid Synthesis of Multifunctional Benzene-1,3,5-tricarboxamides and Creation of Supramolecular Hydrogelators. | Hafeez, S., et al. 2022. J Am Chem Soc. 144: 4057-4070. PMID: 35196454
- Surface-decorated nanoparticles clicked into nanoparticle clusters for oligonucleotide encapsulation. | Mao, W., et al. 2020. RSC Adv. 10: 37040-37049. PMID: 35521231
- One-step synthesis of site-specific antibody-drug conjugates by reprograming IgG glycoengineering with LacNAc-based substrates. | Shi, W., et al. 2022. Acta Pharm Sin B. 12: 2417-2428. PMID: 35646546
- Targeting oncogenic KRAS with molecular brush-conjugated antisense oligonucleotides. | Wang, D., et al. 2022. Proc Natl Acad Sci U S A. 119: e2113180119. PMID: 35858356
- Enzymatic Bioconjugation: A Perspective from the Pharmaceutical Industry. | Debon, A., et al. 2023. JACS Au. 3: 1267-1283. PMID: 37234110
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
3-Azido-1-propanamine, 100 mg | sc-506572 | 100 mg | $109.00 |
