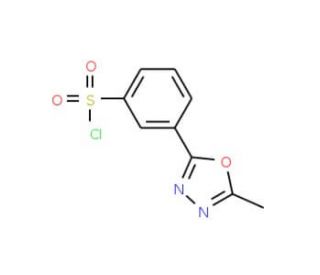
3-(5-Methyl-1,3,4-oxadiazol-2-yl)benzenesulphonyl chloride (CAS 388088-81-1)
QUICK LINKS
3-(5-Methyl-1,3,4-oxadiazol-2-yl)benzenesulfonyl chloride, referred to as MOSC, is a reliable reagent employed in synthetic organic chemistry. As a sulfonyl chloride derivative of 5-methyl-1,3,4-oxadiazole, it serves in diverse synthetic transformations, notably in the creation of an array of heterocyclic compounds. 3-(5-Methyl-1,3,4-oxadiazol-2-yl)benzenesulphonyl chloride finds its use in the production of agrochemicals and a host of specialty chemicals. It′s extensively employed in the synthesis of heterocyclic structures, as well as in the creation of agrochemicals and other specialty chemicals. Beyond these, 3-(5-Methyl-1,3,4-oxadiazol-2-yl)benzenesulphonyl chloride plays a role in the synthesis of various other compounds, including polymers and polysaccharides. It also participates in the synthesis of myriad small molecules, like amino acids, peptides, and nucleotides. Being an electrophilic reagent, 3-(5-Methyl-1,3,4-oxadiazol-2-yl)benzenesulphonyl chloride is capable of interacting with nucleophilic molecules such as amines and alcohols. A common reaction involves the formation of a sulfonamide bond where an oxygen atom replaces the sulfur atom of the sulfonyl chloride. Such reactions generally occur at low temperatures and can be catalyzed using various acids.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
3-(5-Methyl-1,3,4-oxadiazol-2-yl)benzenesulphonyl chloride, 250 mg | sc-260465 | 250 mg | $99.00 |
