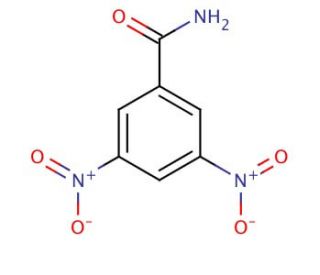
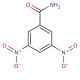
3,5-Dinitrobenzamide (CAS 121-81-3)
QUICK LINKS
3,5-Dinitrobenzamide is a chemical compound that functions as a reagent in organic synthesis. It acts as a precursor in the preparation of various organic compounds, including intermediates and agrochemicals. Its mode of action involves participating in nucleophilic aromatic substitution reactions, allowing for the introduction of functional groups at specific positions on the benzene ring. 3,5-Dinitrobenzamide is utilized as a building block in the synthesis of dyes, pigments, and other specialty chemicals. Its molecular structure enables it to undergo various chemical transformations, making it versatile in the development of new materials and compounds. 3,5-dinitrobenzamide plays a role in the creation of diverse chemical entities with potential applications in different industries.
3,5-Dinitrobenzamide (CAS 121-81-3) References
- Identification of the major urinary and fecal metabolites of 3,5-dinitrobenzamide in chickens and rats. | Shappell, NW., et al. 1999. Chemosphere. 38: 1757-62. PMID: 10101847
- Discrimination of enantiomers of alpha-amino acids by chiral derivatizing reagents from trans-1,2-diaminocyclohexane. | Kaik, M., et al. 2008. Chirality. 20: 301-6. PMID: 17600850
- Mechanism of action and preclinical antitumor activity of the novel hypoxia-activated DNA cross-linking agent PR-104. | Patterson, AV., et al. 2007. Clin Cancer Res. 13: 3922-32. PMID: 17606726
- The nitroreductase prodrug SN 28343 enhances the potency of systemically administered armed oncolytic adenovirus ONYX-411(NTR). | Singleton, DC., et al. 2007. Cancer Gene Ther. 14: 953-67. PMID: 17975564
- Metabolism and excretion of the novel bioreductive prodrug PR-104 in mice, rats, dogs, and humans. | Gu, Y., et al. 2010. Drug Metab Dispos. 38: 498-508. PMID: 20019245
- Decomposition of 3,5-dinitrobenzamide in aqueous solution during UV/H2O2 and UV/TiO2 oxidation processes. | Yan, Y., et al. 2017. Environ Sci Pollut Res Int. 24: 5360-5369. PMID: 28013465
- Design, synthesis and antimycobacterial activity of 3,5-dinitrobenzamide derivatives containing fused ring moieties. | Wang, A., et al. 2018. Bioorg Med Chem Lett. 28: 2945-2948. PMID: 30006066
- Practical screening procedure for chloramphenicol in milk at low parts per billion level. | Schwartz, DP. and McDonough, FE. 1984. J Assoc Off Anal Chem. 67: 563-5. PMID: 6746480
- The metabolic fate of nitromide in the rat. I. Metabolism and excretion. | Facchini, V. and Griffiths, LA. 1980. Xenobiotica. 10: 289-97. PMID: 7415210
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
3,5-Dinitrobenzamide, 10 g | sc-238702 | 10 g | $52.00 |
