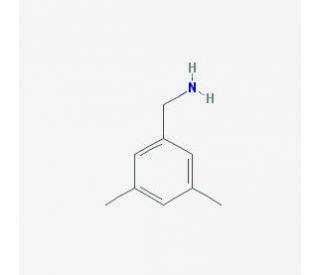
3,5-Dimethylbenzylamine (CAS 78710-55-1)
QUICK LINKS
3,5-Dimethylbenzylamine is a chemical of interest in the field of organic synthesis, where it is often used as a catalyst and an intermediate. In the realm of research, it′s frequently involved in the synthesis of various organic compounds, including biologically active chemicals and agrochemicals, through its role in catalyzing reactions such as Michael additions, Aldol condensations, and Mannich reactions. Its basicity and steric properties make it particularly useful in studies aimed at improving reaction selectivity and yields. Additionally, 3,5-Dimethylbenzylamine′s reactivity with acids, aldehydes, and isocyanates is exploited in the production of amines, amides, and ureas, which are pertinent to material science research, especially in the context of developing new polymeric materials and coatings.
3,5-Dimethylbenzylamine (CAS 78710-55-1) References
- Kinetic versus thermodynamic control during the formation of [2]rotaxanes by a dynamic template-directed clipping process. | Horn, M., et al. 2003. Chemistry. 9: 4046-54. PMID: 12953190
- Structure-activity relationship among purpurinimides and bacteriopurpurinimides: trifluoromethyl substituent enhanced the photosensitizing efficacy. | Gryshuk, A., et al. 2007. J Med Chem. 50: 1754-67. PMID: 17371002
- Highly selective and high-yielding rotaxane synthesis via aminolysis of prerotaxanes consisting of a ring component and a stopper unit. | Hirose, K., et al. 2007. Org Lett. 9: 2969-72. PMID: 17616199
- Preparation and reactions of (beta-trifluoromethyl)vinyl sulfonium salt. | Maeda, R., et al. 2010. Org Lett. 12: 2548-50. PMID: 20455550
- Osmoprotective polymer additives attenuate the membrane pore-forming activity of antimicrobial peptoids. | Smith, PT., et al. 2015. Biopolymers. 103: 227-36. PMID: 25403751
- Synthesis and Biological Evaluation of Fentanyl Analogues Modified at Phenyl Groups with Alkyls. | Qin, Y., et al. 2019. ACS Chem Neurosci. 10: 201-208. PMID: 30179508
- Structure-Activity and Structure-Toxicity Relationships of Peptoid-Based Histone Deacetylase Inhibitors with Dual-Stage Antiplasmodial Activity. | Mackwitz, MKW., et al. 2019. ChemMedChem. 14: 912-926. PMID: 30664827
- Discovery and Structure-Based Optimization of Potent and Selective WD Repeat Domain 5 (WDR5) Inhibitors Containing a Dihydroisoquinolinone Bicyclic Core. | Tian, J., et al. 2020. J Med Chem. 63: 656-675. PMID: 31858797
- Encapsulation of Aromatic Guests in the Bisporphyrin Cavity of a Double-Stranded Spiroborate Helicate: Thermodynamic and Kinetic Studies and the Encapsulation Mechanism. | Ousaka, N., et al. 2021. J Org Chem. 86: 10501-10516. PMID: 34282918
- The chemistry of novolac resins-VI. Reactions between benzoxazine intermediates and model phenols | Zhang, X., & Solomon, D. H. 1998. Polymer. 39(2): 405-412.
- Using ruthenium-catalysed propargylic substitutions for the efficient syntheses of rotaxanes | Tokunaga, Y., Kawai, N., & Shimomura, Y. 2007. Tetrahedron letters. 48(29): 4995-4998.
- Dynamic covalent chemistry of a boronylammonium ion and a crown ether: formation of a C3-symmetric [4] rotaxane | Tokunaga, Y., Ito, T., Sugawara, H., & Nakata, R. 2008. Tetrahedron Letters. 49(21): 3449-3452.
- Hydrothermal Experiments with Protonated Benzylamines Provide Predictions of Temperature-Dependent Deamination Rates for Geochemical Modeling | Robinson, K. J., Gould, I. R., Hartnett, H. E., Williams, L. B., & Shock, E. L. 2021. ACS Earth and Space Chemistry. 5(8): 1997-2012.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
3,5-Dimethylbenzylamine, 1 g | sc-283923 | 1 g | $120.00 | |||
3,5-Dimethylbenzylamine, 5 g | sc-283923A | 5 g | $360.00 |
