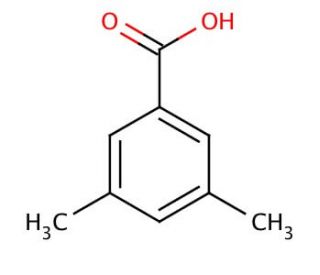
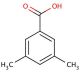
Molecular structure of 3,5-Dimethylbenzoic acid, CAS Number: 499-06-9
3,5-Dimethylbenzoic acid (CAS 499-06-9)
Alternate Names:
3,5-Dimethyl-benzoic Acid; Mesitylenic Acid; NSC 37562
CAS Number:
499-06-9
Molecular Weight:
150.17
Molecular Formula:
C9H10O2
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
3,5-Dimethylbenzoic acid is a reagent with numerous scientific applications. In biochemistry, it serves as a tool for synthesizing a variety of compounds, including amino acids and peptides. Although the exact mechanism of action of 3,5-Dimethylbenzoic acid remains only partially understood, it is believed to function as a proton donor, facilitating the formation of diverse chemical bonds. Additionally, this compound has been identified as a catalyst for specific reactions, such as the hydrolysis of esters and amides.
3,5-Dimethylbenzoic acid (CAS 499-06-9) References
- Organotin(IV) based anti-HCV drugs: synthesis, characterization and biochemical activity. | Shah, FA., et al. 2015. Dalton Trans. 44: 10467-78. PMID: 25978123
- Hemimellitene (1,2,3-trimethylbenzene) in the liver, lung, kidney, and blood, and dimethylbenzoic acid isomers in the liver, lung, kidney and urine of rats after single and repeated inhalation exposure to hemimellitene. | Świercz, R., et al. 2016. Int J Occup Med Environ Health. 29: 113-28. PMID: 26489948
- Diols as Dienophiles: Bridged Carbocycles via Ruthenium(0)-Catalyzed Transfer Hydrogenative Cycloadditions of Cyclohexadiene or Norbornadiene. | Sato, H., et al. 2017. Angew Chem Int Ed Engl. 56: 14667-14671. PMID: 28941001
- Ruthenium(0)-Catalyzed Cycloaddition of 1,2-Diols, Ketols, or Diones via Alcohol-Mediated Hydrogen Transfer. | Sato, H., et al. 2018. Angew Chem Int Ed Engl. 57: 3012-3021. PMID: 29068505
- Design, Synthesis, and Biological Evaluation of Allosteric Effectors That Enhance CO Release from Carboxyhemoglobin. | Goldstein, SR., et al. 2018. ACS Med Chem Lett. 9: 714-718. PMID: 30034606
- One-pot, multi-component synthesis and structure-activity relationships of peptoid-based histone deacetylase (HDAC) inhibitors targeting malaria parasites. | Diedrich, D., et al. 2018. Eur J Med Chem. 158: 801-813. PMID: 30245402
- Catalytic Hydrothiolation: Counterion-Controlled Regioselectivity. | Yang, XH., et al. 2019. J Am Chem Soc. 141: 3006-3013. PMID: 30735362
- Multicomponent Synthesis, Binding Mode, and Structure-Activity Relationship of Selective Histone Deacetylase 6 (HDAC6) Inhibitors with Bifurcated Capping Groups. | Reßing, N., et al. 2020. J Med Chem. 63: 10339-10351. PMID: 32803970
- N-Aminopyridinium Ylide-Directed, Copper-Promoted Chalcogenation of Arene C-H Bonds. | Nguyen, H. and Daugulis, O. 2020. J Org Chem. 85: 13069-13079. PMID: 33000944
- Rational design of selective inhibitors of PARP4. | Kirby, IT., et al. 2021. RSC Med Chem. 12: 1950-1957. PMID: 34825190
- Optimization of a Pyrimidinone Series for Selective Inhibition of Ca2+/Calmodulin-Stimulated Adenylyl Cyclase 1 Activity for the Treatment of Chronic Pain. | Scott, JA., et al. 2022. J Med Chem. 65: 4667-4686. PMID: 35271288
- Data of chemical composition of the particles from OH-initiated oxidation of 1,3,5-trimethylbenzene. | Lin, X., et al. 2022. Data Brief. 42: 108152. PMID: 35496493
- Chemoselective derivatisation and ultrahigh resolution mass spectrometry for the determination of hydroxyl functional groups within complex bio-oils. | Palacio Lozano, DC., et al. 2023. RSC Adv. 13: 17727-17741. PMID: 37312997
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
3,5-Dimethylbenzoic acid, 25 g | sc-256594 | 25 g | $65.00 |
