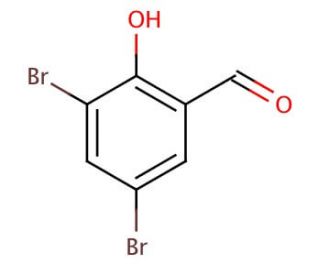
3,5-Dibromosalicylaldehyde (CAS 90-59-5)
See product citations (1)
QUICK LINKS
3,5-Dibromosalicylaldehyde, also referred to as 3,5-DBS, is an aromatic aldehyde that has garnered significant attention in the chemical industry due to its potential applications. Scientists have extensively employed 3,5-Dibromosalicylaldehyde in various scientific research fields. It has found utility in the synthesis of diverse heterocyclic compounds, such as indoles and quinolines, serving as a valuable reagent for the synthesis of biologically active compounds. Its application extends to the synthesis of polysaccharides, other polymers, and polycarbonates. Understanding the intricate mechanism of action of 3,5-Dibromosalicylaldehyde remains an ongoing endeavor. It is currently hypothesized that the presence of bromine atoms in the molecule renders it highly reactive due to their electron-withdrawing nature. This increased reactivity allows 3,5-DBS to engage in diverse reactions with an array of compounds, including proteins and other vital biological molecules.
3,5-Dibromosalicylaldehyde (CAS 90-59-5) References
- Deuterium isotope effect on 13C chemical shifts of tetrabutylammonium salts of Schiff bases amino acids. | Rozwadowski, Z. 2006. Magn Reson Chem. 44: 881-6. PMID: 16741983
- Synthesis, structures and urease inhibition studies of Schiff base metal complexes derived from 3,5-dibromosalicylaldehyde. | Cui, Y., et al. 2012. Eur J Med Chem. 58: 323-31. PMID: 23142672
- Tick-borne encephalitis virus triggers inositol-requiring enzyme 1 (IRE1) and transcription factor 6 (ATF6) pathways of unfolded protein response. | Yu, C., et al. 2013. Virus Res. 178: 471-7. PMID: 24177270
- Japanese encephalitis virus induces apoptosis by the IRE1/JNK pathway of ER stress response in BHK-21 cells. | Huang, M., et al. 2016. Arch Virol. 161: 699-703. PMID: 26660165
- Pre-ischemia melatonin treatment alleviated acute neuronal injury after ischemic stroke by inhibiting endoplasmic reticulum stress-dependent autophagy via PERK and IRE1 signalings. | Feng, D., et al. 2017. J Pineal Res. 62: PMID: 28178380
- A new azine derivative colorimetric and fluorescent dual-channel probe for cyanide detection. | Yu, B., et al. 2017. Spectrochim Acta A Mol Biomol Spectrosc. 184: 249-254. PMID: 28505606
- RACK1 upregulation induces neuroprotection by activating the IRE1-XBP1 signaling pathway following traumatic brain injury in rats. | Ni, H., et al. 2018. Exp Neurol. 304: 102-113. PMID: 29518365
- Homer1a Attenuates Endoplasmic Reticulum Stress-Induced Mitochondrial Stress After Ischemic Reperfusion Injury by Inhibiting the PERK Pathway. | Wei, J., et al. 2019. Front Cell Neurosci. 13: 101. PMID: 30930751
- The effect of halogenation of salicylaldehyde on the antiproliferative activities of {Δ/Λ-[Ru(bpy)2(X,Y-sal)]BF4} complexes. | Taghizadeh Shool, M., et al. 2022. Dalton Trans. 51: 7658-7672. PMID: 35510940
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
3,5-Dibromosalicylaldehyde, 10 g | sc-232077 | 10 g | $56.00 |
