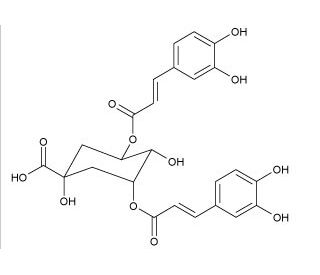
Molecular structure of 3,5-Di-O-caffeoylquinic acid, CAS Number: 2450-53-5
3,5-Di-O-caffeoylquinic acid (CAS 2450-53-5)
Alternate Names:
Isochlorogenic acid
Application:
3,5-Di-O-caffeoylquinic acid is an antioxidant that has been shown to have antiproliferative activity
CAS Number:
2450-53-5
Purity:
≥97%
Molecular Weight:
516.5
Molecular Formula:
C25H24O12
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
3,5-Di-O-caffeoylquinic acid, a dicaffeoylquinic acid derivative, plays a significant role in the realm of natural product chemistry and plant-based research. This compound, characterized by the esterification of quinic acid with two molecules of caffeic acid at the 3 and 5 positions, is notable for its involvement in plant defense mechanisms and stress responses. In scientific studies, it is frequently employed to investigate its antioxidant, anti-inflammatory, and UV-protection properties at a cellular level, focusing on its ability to scavenge free radicals and modulate signaling pathways related to oxidative stress and inflammation.
3,5-Di-O-caffeoylquinic acid (CAS 2450-53-5) References
- Angiotensin converting enzyme inhibitors from Cuscuta japonica Choisy. | Oh, H., et al. 2002. J Ethnopharmacol. 83: 105-8. PMID: 12413714
- In vitro antioxidative effects and tyrosinase inhibitory activities of seven hydroxycinnamoyl derivatives in green coffee beans. | Iwai, K., et al. 2004. J Agric Food Chem. 52: 4893-8. PMID: 15264931
- Changes in caffeic acid derivatives in sweet potato (Ipomoea batatas L.) during cooking and processing. | Takenaka, M., et al. 2006. Biosci Biotechnol Biochem. 70: 172-7. PMID: 16428835
- Neuroprotective effect of 3,5-di-O-caffeoylquinic acid on SH-SY5Y cells and senescence-accelerated-prone mice 8 through the up-regulation of phosphoglycerate kinase-1. | Han, J., et al. 2010. Neuroscience. 169: 1039-45. PMID: 20570715
- Structure-activity relationship of caffeoylquinic acids on the accelerating activity on ATP production. | Miyamae, Y., et al. 2011. Chem Pharm Bull (Tokyo). 59: 502-7. PMID: 21467684
- Neuroprotective effects of 3,5-di-o-caffeoylquinic acid in vitro and in vivo. | Han, J. and Isoda, H. 2011. BMC Proc. 5 Suppl 8: P20. PMID: 22373134
- [Molecular docking of chlorogenic acid, 3,4-di-O-caffeoylquinic acid and 3,5-di-O-caffeoylquinic acid with human serum albumin]. | Zhou, J., et al. 2012. Zhong Xi Yi Jie He Xue Bao. 10: 1149-54. PMID: 23073199
- Gymnaster koraiensis and its major components, 3,5-di-O-caffeoylquinic acid and gymnasterkoreayne B, reduce oxidative damage induced by tert-butyl hydroperoxide or acetaminophen in HepG2 cells. | Jho, EH., et al. 2013. BMB Rep. 46: 513-8. PMID: 24148773
- NMR Studies of Hetero-Association of Caffeine with di-O-Caffeoylquinic Acid Isomers in Aqueous Solution. | D'Amelio, N., et al. 2015. Food Biophys. 10: 235-243. PMID: 26213521
- Jasmonates promote enhanced production of bioactive caffeoylquinic acid derivative in Eclipta prostrata (L.) L. hairy roots. | Maciel, G., et al. 2022. Plant Cell Tissue Organ Cult. 149: 363-369. PMID: 34840370
- Petasites japonicus extract exerts anti-malarial effects by inhibiting platelet activation. | Yun, HS., et al. 2022. Phytomedicine. 102: 154167. PMID: 35598522
- Biosynthetic studies through feeding experiments in Eclipta prostrata (L.) L. hairy roots. | Lopes, AA., et al. 2022. Plant Cell Tissue Organ Cult. 151: 215-219. PMID: 35875188
Inhibitor of:
INTS4.Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
3,5-Di-O-caffeoylquinic acid, 1 mg | sc-202418 | 1 mg | $156.00 |
