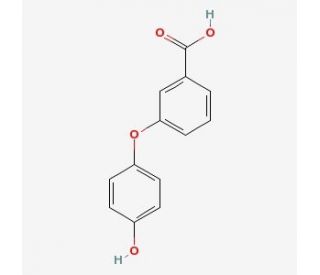
Molecular structure of 3-(4′-Hydroxy)phenoxybenzoic Acid, CAS Number: 35065-12-4
3-(4′-Hydroxy)phenoxybenzoic Acid (CAS 35065-12-4)
Alternate Names:
4′-hydroxy-3-phenoxybenzoic acid
Application:
3-(4′-Hydroxy)phenoxybenzoic Acid is a metabolite of pyrethroid insecticides
CAS Number:
35065-12-4
Purity:
≥98%
Molecular Weight:
230.22
Molecular Formula:
C13H10O4
Supplemental Information:
This is classified as a Dangerous Good for transport and may be subject to additional shipping charges.
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
3-(4′-Hydroxy)phenoxybenzoic Acid is a metabolite of pyrethroid insecticides such as Permethrin, Deltamethrin, Cypermethrin, Fenvalerate, and Decamethrin.
3-(4′-Hydroxy)phenoxybenzoic Acid (CAS 35065-12-4) References
- Lack of significant estrogenic or antiestrogenic activity of pyrethroid insecticides in three in vitro assays based on classic estrogen receptor alpha-mediated mechanisms. | Saito, K., et al. 2000. Toxicol Sci. 57: 54-60. PMID: 10966511
- Structure-activity relationships for a large diverse set of natural, synthetic, and environmental estrogens. | Fang, H., et al. 2001. Chem Res Toxicol. 14: 280-94. PMID: 11258977
- Effects of currently used pesticides in assays for estrogenicity, androgenicity, and aromatase activity in vitro. | Andersen, HR., et al. 2002. Toxicol Appl Pharmacol. 179: 1-12. PMID: 11884232
- Diphenyl ether cleavage of 3-phenoxybenzoic acid by chicken kidney microsomal preparations. | Akhtar, MH. and Mahadevan, S. 1992. Drug Metab Dispos. 20: 356-9. PMID: 1355708
- The metabolism of cypermethrin in man: differences in urinary metabolite profiles following oral and dermal administration. | Woollen, BH., et al. 1992. Xenobiotica. 22: 983-91. PMID: 1413886
- Worker exposure to, and absorption of, cypermethrin during aerial application of an 'ultra low volume' formulation to cotton. | Chester, G., et al. 1987. Arch Environ Contam Toxicol. 16: 69-78. PMID: 3813658
- Metabolism of phenothrin or 3-phenoxybenzyl d-trans-chrysanthemumate in mammals | Miyamoto, J., Suzuki, T., & Nakae, C. 1974. Pesticide biochemistry and physiology. 4(4): 438-450.
- In vitro studies of the metabolism of atrazine, simazine, and terbutryn in several vertebrate species | Adams, N. H., Levi, P. E., & Hodgson, E. 1990. Journal of agricultural and food chemistry. 38(6): 1411-1417.
- Mechanism of synergism between the pyrethroid insecticide λ-cyhalothrin and the imidazole fungicide prochloraz, in the honeybee (Apis mellifera L.) | Pilling, E. D., Bromleychallenor, K. A. C., Walker, C. H., & Jepson, P. C. 1995. Pesticide biochemistry and physiology. 51(1): 1-11.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
3-(4′-Hydroxy)phenoxybenzoic Acid, 10 mg | sc-209468 | 10 mg | $300.00 |
