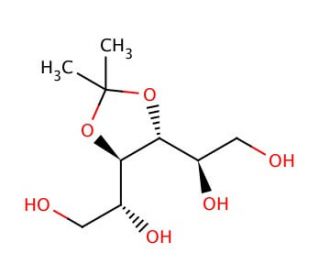
3,4-O-Isopropylidene-D-mannitol (CAS 3969-84-4)
QUICK LINKS
3,4-O-Isopropylidene-D-mannitol is a critical intermediate in carbohydrate chemistry, used extensively in the synthesis and study of sugar derivatives and polyols. This compound features an isopropylidene group protecting the 3 and 4 positions of D-mannitol, providing stability and selectivity in synthetic transformations. The protection of these hydroxyl groups is essential for controlling the reactivity of the remaining free hydroxyls, enabling precise modifications necessary for the creation of complex carbohydrate structures. In research, 3,4-O-Isopropylidene-D-mannitol is employed to investigate the mechanisms of glycosylation and deprotection processes, which are fundamental to understanding carbohydrate metabolism and biosynthesis. This compound serves as a building block in the preparation of various glycosides, oligosaccharides, and glycoconjugates, facilitating studies on enzyme activity, particularly glycosyltransferases and glycosidases. By providing a protected intermediate, it allows researchers to perform regioselective reactions, ensuring that modifications occur at desired positions on the sugar molecule. Through these applications, it significantly contributes to the advancement of glycoscience, providing insights into the complex mechanisms governing carbohydrate-related biochemical pathways.
3,4-O-Isopropylidene-D-mannitol (CAS 3969-84-4) References
- Synthesis of chiral hydroxyl phospholanes from D-mannitol and their use in asymmetric catalytic reactions. | Li, W., et al. 2000. J Org Chem. 65: 3489-96. PMID: 10843636
- Polyhydroxylated azepanes as new motifs for DNA minor groove binding agents. | Johnson, HA. and Thomas, NR. 2002. Bioorg Med Chem Lett. 12: 237-41. PMID: 11755363
- Zeolite catalyzed selective deprotection of di- and tri-O-isopropylidene sugar acetals. | Bhaskar, PM., et al. 2008. Carbohydr Res. 343: 1801-7. PMID: 18502410
- Cytotoxicity Study on Luminescent Nanocrystals Containing Phospholipid Micelles in Primary Cultures of Rat Astrocytes. | Latronico, T., et al. 2016. PLoS One. 11: e0153451. PMID: 27097043
- Synthesis and Biological Evaluation of Trehalose-based Bi-aryl Derivatives as C-type Lectin Ligands. | Rasheed, OK., et al. 2023. Tetrahedron. 132: PMID: 36874612
- Synthesis of new sugar derivatives having potential antitumour activity: Part IX. Cyclic phosphoric acid derivatives. | Kuszmann, J. and L. Vargha. 1966. Carbohydrate Research. 3.1: 38-46.
- Some reactions of 1, 6-di-Op-tolylsulphonyl-D-mannitol and its derivatives. | Kuszmann, Jànos and Pál Sohár. 1974. Carbohydrate Research. 35.1: 97-102.
- Convenient Synthesis of the Protein Thermal‐Stabilizer Mannosylglycerate. | Cheikh, Khaled El, et al. 2016. ChemistrySelect. 1.10: 2471-2473.
- Tunable hydrophilicity of redox D-mannitol-based polyurethanes modulates glutathione response. | Romero-Azogil, Lucía, et al. 2017. European Polymer Journal. 94: 259-269.
- Hydrolytic degradation of D-mannitol-based polyurethanes. | Romero-Azogil, Lucía, et al. 2018. Polymer degradation and stability. 153: 262-271.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
3,4-O-Isopropylidene-D-mannitol, 5 g | sc-256586 | 5 g | $210.00 |
