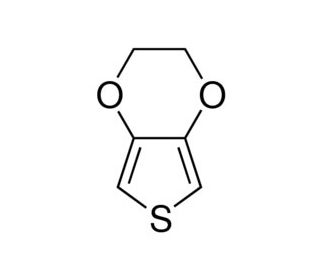
Molecular structure of 3,4-Ethylenedioxythiophene, CAS Number: 126213-50-1
3,4-Ethylenedioxythiophene (CAS 126213-50-1)
Alternate Names:
2,3-Dihydrothieno[3,4-β]-1,4-dioxin
Application:
3,4-Ethylenedioxythiophene is a monomer used to synthesize the conductive polymer PEDT
CAS Number:
126213-50-1
Molecular Weight:
142.18
Molecular Formula:
C6H6O2S
Supplemental Information:
This is classified as a Dangerous Good for transport and may be subject to additional shipping charges.
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
3,4-Ethylenedioxythiophene (EDOT) is a monomer in the synthesis of poly(3,4-ethylenedioxythiophene), abbreviated as PEDT or PEDOT, a highly conductive polymer that has garnered significant attention in various fields of science and technology. 3,4-Ethylenedioxythiophene itself features a thiophene ring—a sulfur-containing heterocycle—bridged by an ethylene dioxy group, which imparts the molecule with unique electronic properties conducive to polymerization. The polymerization of 3,4-Ethylenedioxythiophene, often achieved through oxidative processes, leads to the formation of PEDOT, a polymer distinguished by its excellent electrical conductivity and stability under oxidative conditions.
3,4-Ethylenedioxythiophene (CAS 126213-50-1) References
- Long-chain 3,4-ethylenedioxythiophene/thiophene oligomers and semiconducting thin films prepared by their electropolymerization. | de Silva, KM., et al. 2012. ACS Appl Mater Interfaces. 4: 5430-41. PMID: 22970915
- 3,4-Ethylenedioxythiophene in planarizable push-pull oligothiophenes. | Dal Molin, M. and Matile, S. 2013. Org Biomol Chem. 11: 1952-7. PMID: 23389656
- A simple 3,4-ethylenedioxythiophene based hole-transporting material for perovskite solar cells. | Li, H., et al. 2014. Angew Chem Int Ed Engl. 53: 4085-8. PMID: 24634079
- Synthesis and structure-activity relationship of amidine derivatives of 3,4-ethylenedioxythiophene as novel antibacterial agents. | Stolić, I., et al. 2015. Eur J Med Chem. 90: 68-81. PMID: 25461312
- Conductive Textiles via Vapor-Phase Polymerization of 3,4-Ethylenedioxythiophene. | Ala, O., et al. 2017. ACS Appl Mater Interfaces. 9: 29038-29046. PMID: 28758733
- Copolymers Based on 1,3-Bis(carbazol-9-yl)benzene and Three 3,4-Ethylenedioxythiophene Derivatives as Potential Anodically Coloring Copolymers in High-Contrast Electrochromic Devices. | Kuo, CW., et al. 2016. Polymers (Basel). 8: PMID: 30974647
- Donor-Acceptor-Donor Copolymers with 3,4-Ethylenedioxythiophene Moiety: Electropolymerization and Effect on Optoelectronic and Electrochromic Properties. | Singhal, S., et al. 2019. ACS Omega. 4: 3484-3492. PMID: 31459563
- Poly(3,4-ethylenedioxythiophene)/Te/Single-Walled Carbon Nanotube Composites with High Thermoelectric Performance Promoted by Electropolymerization. | Yin, S., et al. 2020. ACS Appl Mater Interfaces. 12: 3547-3553. PMID: 31887003
- Solution-Processable Copolymers Based on Triphenylamine and 3,4-Ethylenedioxythiophene: Facile Synthesis and Multielectrochromism. | Neo, WT., et al. 2020. Macromol Rapid Commun. 41: e2000156. PMID: 32578302
- Boron difluoride formazanates with thiophene and 3,4-ethylenedioxythiophene capping and their electrochemical polymerization. | Kumar, C., et al. 2020. Dalton Trans. 49: 13202-13206. PMID: 32966457
- Physical and Electrochemical Properties of Soluble 3,4-Ethylenedioxythiophene (EDOT)-Based Copolymers Synthesized via Direct (Hetero)Arylation Polymerization. | Guo, Q., et al. 2021. Front Chem. 9: 753840. PMID: 34778210
- Biguanide- and Oligo(Ethylene Glycol)-Functionalized Poly(3,4-Ethylenedioxythiophene): Electroactive, Antimicrobial, and Antifouling Surface Coatings. | Ayalew, H., et al. 2022. Front Chem. 10: 955260. PMID: 35991598
- Electroactive and Stretchable Hydrogels of 3,4-Ethylenedioxythiophene/thiophene Copolymers. | Chen, PW., et al. 2023. ACS Omega. 8: 6753-6761. PMID: 36844572
- AC-Bipolar Electropolymerization of 3,4-Ethylenedioxythiophene in Ionic Liquids. | Chen, Z., et al. 2023. Langmuir. 39: 4450-4455. PMID: 36919992
- Inclusion Complexes of 3,4-Ethylenedioxythiophene with Per-Modified β- and γ-Cyclodextrins. | Farcas, A., et al. 2023. Molecules. 28: PMID: 37110637
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
3,4-Ethylenedioxythiophene, 5 g | sc-475384 | 5 g | $26.00 | |||
3,4-Ethylenedioxythiophene, 25 g | sc-475384A | 25 g | $109.00 | |||
3,4-Ethylenedioxythiophene, 100 g | sc-475384B | 100 g | $379.00 | |||
3,4-Ethylenedioxythiophene, 250 g | sc-475384C | 250 g | $900.00 |
