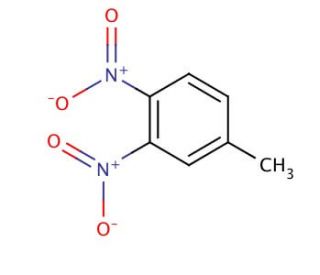
Molecular structure of 3,4-Dinitrotoluene, CAS Number: 610-39-9
3,4-Dinitrotoluene (CAS 610-39-9)
Application:
3,4-Dinitrotoluene is a useful standard used in scientific research
CAS Number:
610-39-9
Molecular Weight:
182.13
Molecular Formula:
C7H6N2O4
Supplemental Information:
This is classified as a Dangerous Good for transport and may be subject to additional shipping charges.
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
3,4-Dinitrotoluene is used in scientific research as a staple reagent for crafting nitroaromatic compounds, nitroalkanes, and nitroheterocycles. It is further utilized as a standard for environmental analysis. It also serves as a model compound for the investigation of the environmental impact of nitroaromatics due to its persistence and presence in industrial waste. It is understood to undergo metabolism, with its byproducts potentially disrupting enzymatic activities, leading to various toxicological outcomes.
3,4-Dinitrotoluene (CAS 610-39-9) References
- The response of adult rat sertoli cells, immortalized by a temperature-sensitive mutant of SV40, to 1,2-dinitrobenzene, 1,3-dinitrobenzene, 2,4-dinitrotoluene, 3,4-dinitrotoluene, and cadmium. | Sorenson, DR. and Brabec, M. 2003. Cell Biol Toxicol. 19: 107-19. PMID: 12776928
- Anaerobic biotransformation of dinitrotoluene isomers by Lactococcus lactis subsp. lactis strain 27 isolated from earthworm intestine. | Shin, KH., et al. 2005. Chemosphere. 61: 30-9. PMID: 16157167
- Gas chromatography positive chemical ionization and tandem mass spectrometry for the analysis of organic high explosives. | Perr, JM., et al. 2005. Talanta. 67: 430-6. PMID: 18970185
- Structure-activity relationship for the intrinsic hepatotoxicity of dinitrotoluenes. | Spanggord, RJ., et al. 1990. Chem Res Toxicol. 3: 551-8. PMID: 2103326
- Evaluation of the dependence of aqueous solubility of nitro compounds on temperature and salinity: a COSMO-RS simulation. | Kholod, YA., et al. 2011. Chemosphere. 83: 287-94. PMID: 21215986
- Effects of dinitrotoluenes on morphological cell transformation and intercellular communication in Syrian hamster embryo cells. | Holen, I., et al. 1990. J Toxicol Environ Health. 29: 89-98. PMID: 2299690
- Dissociation Enthalpies of Chloride Adducts of Nitrate and Nitrite Explosives Determined by Ion Mobility Spectrometry. | Rajapakse, MY., et al. 2016. J Phys Chem A. 120: 690-8. PMID: 26777731
- Tissue distribution of 2,4-dinitrotoluene and its metabolites in male and female Fischer-344 rats. | Rickert, DE. and Long, RM. 1980. Toxicol Appl Pharmacol. 56: 286-93. PMID: 7466827
- Phototoxicology. 1. Light-enhanced toxicity of TNT and some related compounds to Daphnia magna and Lytechinus variagatus embryos. | Davenport, R., et al. 1994. Ecotoxicol Environ Saf. 27: 14-22. PMID: 7525201
- Optimization of the Flow Rate in High Pressure Liquid Chromatography of explosives | Kaiser, M. 1996. Propellants, explosives, pyrotechnics. 21(5): 266-270.
- Design and evaluation of a novel hemispherical FAIMS cell | Jennifer G. Bryant, Marilyn Prieto, Todd A. Prox, Richard A. Yost. 2010. International Journal of Mass Spectrometry. 298(1–3): 41-44.
- Lanthanide-organic frameworks based on terphenyl-tetracarboxylate ligands: syntheses, structures, optical properties and selective sensing of nitro explosives | Zhou, X., Zhu, Y., Li, L., Yang, T., Wang, J., & Huang, W. 2017. Science China Chemistry. 60: 1130-1135.
- Effective Removal of Nitrotoluene Compounds from Aqueous Solution Using Magnetic-Activated Carbon Nanocomposites (m-Fe3O4@ACCs) | Sovizi, M. R., & Esmaeilzade, A. 2018. Russian Journal of Applied Chemistry. 91: 253-263.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
3,4-Dinitrotoluene, 500 mg | sc-232030 | 500 mg | $275.00 |
