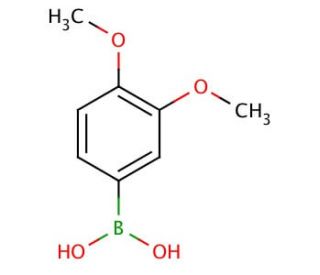
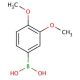
3,4-Dimethoxyphenylboronic acid (CAS 122775-35-3)
QUICK LINKS
3,4-Dimethoxyphenylboronic acid functions as a key reagent in organic synthesis. It acts as a versatile building block for the construction of various biaryl compounds. 3,4-Dimethoxyphenylboronic Acid participates in Suzuki-Miyaura cross-coupling reactions, where it forms a boronate ester intermediate that undergoes transmetalation with an aryl halide, leading to the formation of a new carbon-carbon bond. Through this mechanism, 3,4-Dimethoxyphenylboronic acid enables the efficient and selective formation of complex organic molecules, making it useful for the synthesis of diverse chemical compounds in research and development applications. Its mechanism of action involves the activation of the boron atom, facilitating its coupling with aryl halides to generate biaryl products with high regio- and stereo-selectivity.
3,4-Dimethoxyphenylboronic acid (CAS 122775-35-3) References
- Selective Inhibitors of Cyclin G Associated Kinase (GAK) as Anti-Hepatitis C Agents. | Kovackova, S., et al. 2015. J Med Chem. 58: 3393-410. PMID: 25822739
- Isothiazolo[4,3-b]pyridines as inhibitors of cyclin G associated kinase : synthesis, structure-activity relationship studies and antiviral activity. | Li, J., et al. 2015. Medchemcomm. 6: 1666-1672. PMID: 26925208
- Imidazo[1,2-a]quinoxalines Derivatives Grafted with Amino Acids: Synthesis and Evaluation on A375 Melanoma Cells. | Chouchou, A., et al. 2018. Molecules. 23: PMID: 30445763
- Synthesis and Structure-Activity Relationships of 3,5-Disubstituted-pyrrolo[2,3- b]pyridines as Inhibitors of Adaptor-Associated Kinase 1 with Antiviral Activity. | Verdonck, S., et al. 2019. J Med Chem. 62: 5810-5831. PMID: 31136173
- Novel Human Neutral Sphingomyelinase 2 Inhibitors as Potential Therapeutics for Alzheimer's Disease. | Šála, M., et al. 2020. J Med Chem. 63: 6028-6056. PMID: 32298582
- Discovery of Potent and Orally Bioavailable Small Molecule Antagonists of Toll-like Receptors 7/8/9 (TLR7/8/9). | Mussari, CP., et al. 2020. ACS Med Chem Lett. 11: 1751-1758. PMID: 32944143
- Catalytic enantioselective arylative cyclizations of alkynyl 1,3-diketones by 1,4-rhodium(i) migration. | Groves, A., et al. 2020. Chem Sci. 11: 2759-2764. PMID: 34084335
- Synthesis of new substituted 7,12-dihydro-6,12-methanodibenzo[c,f]azocine-5-carboxylic acids containing a tetracyclic tetrahydroisoquinoline core structure. | Grajewska, A., et al. 2021. Beilstein J Org Chem. 17: 2511-2519. PMID: 34691267
- A Phenylfurocoumarin Derivative Reverses ABCG2-Mediated Multidrug Resistance In Vitro and In Vivo. | Kokubo, S., et al. 2021. Int J Mol Sci. 22: PMID: 34830383
- 7H-Pyrrolo[2,3-d]pyrimidine-4-amines as Potential Inhibitors of Plasmodium falciparum Calcium-Dependent Protein Kinases. | Seanego, TD., et al. 2022. ChemMedChem. 17: e202200421. PMID: 36106757
- Important Requirements for Desorption/Ionization Mass Spectrometric Measurements of Temozolomide-Induced 2'-Deoxyguanosine Methylations in DNA. | Fresnais, M., et al. 2023. Cancers (Basel). 15: PMID: 36765673
- A Single-Component Molecular Glass Resist Based on Tetraphenylsilane Derivatives for Electron Beam Lithography. | Wang, Y., et al. 2023. ACS Omega. 8: 12173-12182. PMID: 37033792
- Diastereoselective Synthesis of (-)-6,7-Dimethoxy-1,2,3,4-tetrahydroisoquinoline-1-carboxylic Acid via Morpholinone Derivatives. | Chrzanowska, M., et al. 2023. Molecules. 28: PMID: 37049962
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
3,4-Dimethoxyphenylboronic acid, 5 g | sc-254537 | 5 g | $50.00 |
