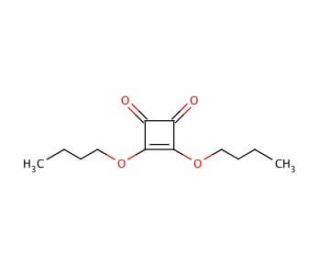
3,4-Dibutoxy-3-cyclobutene-1,2-dione (CAS 2892-62-8)
QUICK LINKS
3,4-Dibutoxy-3-cyclobutene-1,2-dione, referred to as DBCD, is a cyclic diketone compound, characterized by two ketone groups linked within a cyclic ring structure. 3,4-Dibutoxy-3-cyclobutene-1,2-dione plays a significant role as an intermediate in the synthesis of diverse organic compounds, making it a valuable component in various scientific research applications. In scientific research, 3,4-Dibutoxy-3-cyclobutene-1,2-dione has demonstrated its versatility in numerous applications. Notably, it serves as a substrate in the synthesis of diverse organic compounds, including heterocycles, polymers, and pharmaceuticals. Moreover, its catalytic properties have proven effective in promoting the synthesis of cyclic compounds and various complex organic molecules. Furthermore, 3,4-Dibutoxy-3-cyclobutene-1,2-dione has proven instrumental as an intermediate in the synthesis of natural products such as steroids and terpenes, further accentuating its importance in the realm of organic chemistry. The mechanism of action for 3,4-Dibutoxy-3-cyclobutene-1,2-dione is intricately tied to the specific reaction it is involved in. For instance, in the Wittig reaction, the phosphonium ylide reacts with aldehydes or ketones, ultimately forming an alkene. Meanwhile, in the Stille reaction, the organostannane reacts with aldehydes or ketones, leading to the formation of alkyl-substituted alkenes. Lastly, in the Horner-Wadsworth-Emmons reaction, the phosphonate ester reacts with aldehydes or ketones, resulting in the formation of an alkene. Throughout these reactions, the catalytic base plays a pivotal role in deprotonating the reactants and facilitating the reaction process.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
3,4-Dibutoxy-3-cyclobutene-1,2-dione, 5 g | sc-231977 | 5 g | $73.00 |
