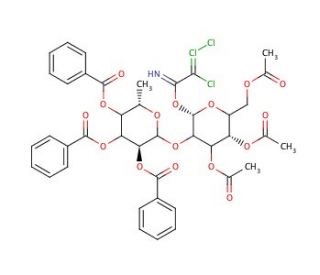
3,4,6-Tri-O-acetyl-2-O-(2′,3′,4′-tri-O-benzoyl-α-L-fucopyranosyl)-galactopyranosyl Trichloroacetimidate
QUICK LINKS
3,4,6-Tri-O-acetyl-2-O-(2′,3′,4′-tri-O-benzoyl-α-L-fucopyranosyl)-galactopyranosyl trichloroacetimidate, a highly functionalized carbohydrate derivative, plays a pivotal role in carbohydrate chemistry research. Its unique structure makes it a versatile building block for synthesizing complex oligosaccharides and glycoconjugates. In research, this compound serves as a glycosyl donor in glycosylation reactions, facilitating the formation of glycosidic bonds between sugar units. The trichloroacetimidate moiety enhances the reactivity of the glycosyl donor, enabling efficient coupling with various acceptor molecules under mild conditions. Chemists utilize this compound to construct diverse glycostructures with precise stereochemistry and regioselectivity, mimicking natural carbohydrate motifs found in biological systems. Moreover, 3,4,6-Tri-O-acetyl-2-O-(2′,3′,4′-tri-O-benzoyl-α-L-fucopyranosyl)-galactopyranosyl trichloroacetimidate has been instrumental in the synthesis of glycoconjugate vaccines, glycopeptides, and glycolipids, contributing to the development of novel agents and diagnostic tools. Its applications extend to glycobiology studies, where researchers investigate carbohydrate-mediated interactions, such as cell-cell recognition, host-pathogen interactions, and immune responses. Overall, this compound serves as a valuable tool for elucidating the roles of carbohydrates in biological processes and advancing various fields of research, including chemistry, biochemistry, and molecular biology.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
3,4,6-Tri-O-acetyl-2-O-(2',3',4'-tri-O-benzoyl-α-L-fucopyranosyl)-galactopyranosyl Trichloroacetimidate, 5 mg | sc-223567 | 5 mg | $320.00 |
