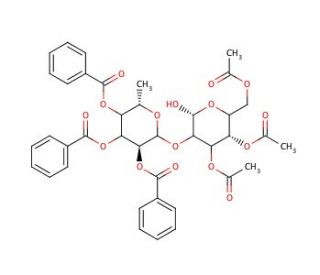
3,4,6-Tri-O-acetyl-2-O-(2′,3′,4′-tri-O-benzoyl-α-L-fucopyranosyl)-galactopyranose
QUICK LINKS
3,4,6-Tri-O-acetyl-2-O-(2′,3′,4′-tri-O-benzoyl-α-L-fucopyranosyl)-galactopyranose is a key compound utilized in carbohydrate chemistry research due to its role in the synthesis and modification of complex oligosaccharides. The chemical mechanism involves the protection of specific hydroxyl groups on both the fucopyranosyl and galactopyranose moieties, facilitating selective glycosylation reactions and enabling the construction of complex sugar structures. Researchers often employ this compound to investigate the structural and functional properties of carbohydrates, including their interactions with proteins, enzymes, and other biomolecules. Additionally, 3,4,6-Tri-O-acetyl-2-O-(2′,3′,4′-tri-O-benzoyl-α-L-fucopyranosyl)-galactopyranose has been instrumental in the development of glycan microarrays, glycoconjugates, and glycosidic linkages, which are vital tools for studying glycobiology, cell signaling, and immune response modulation. Its application extends to various fields such as chemical biology, glycobiotechnology, and drug discovery, where a deeper understanding of carbohydrate structures and functions is essential for advancing research in diverse areas of science and technology. Overall, this chemical plays a crucial role in elucidating the complex roles of carbohydrates in biological processes and their potential applications in various research endeavors.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
3,4,6-Tri-O-acetyl-2-O-(2',3',4'-tri-O-benzoyl-α-L-fucopyranosyl)-galactopyranose, 5 mg | sc-223566 | 5 mg | $300.00 |
