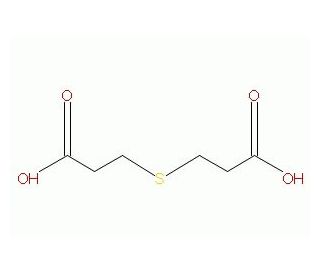
3,3′-Thiodipropionic acid (CAS 111-17-1)
QUICK LINKS
3,3′-Thiodipropionic acid is a compound that functions as a stabilizer and antioxidant in various experiments. It acts by inhibiting the oxidation of other substances, thereby the degradation of materials and maintaining their integrity. At the molecular level, interacts with free radicals and reactive oxygen species, neutralizing their harmful effects and preserving the quality of the experimental samples. By disrupting the chain reaction of oxidation, it effectively extends the shelf life and stability of the materials under investigation. The mode of action involves the donation of hydrogen atoms to free radicals, forming stable products and interrupting the propagation of oxidative reactions. In this way, 3,3′-Thiodipropionic acid plays a functional role in maintaining the reliability and consistency of experimental results by protecting the integrity of the substances involved.
3,3′-Thiodipropionic acid (CAS 111-17-1) References
- Biosynthesis and biodegradability of copolythioesters from 3,3'-thiodipropionic acid and plant oils by Cupriviadus necator. | Kamei, Y., et al. 2007. Macromol Biosci. 7: 364-72. PMID: 17370275
- 3-mercaptopropionate dioxygenase, a cysteine dioxygenase homologue, catalyzes the initial step of 3-mercaptopropionate catabolism in the 3,3-thiodipropionic acid-degrading bacterium variovorax paradoxus. | Bruland, N., et al. 2009. J Biol Chem. 284: 660-672. PMID: 19001372
- Biodegradation of the xenobiotic organic disulphide 4,4'-dithiodibutyric acid by Rhodococcus erythropolis strain MI2 and comparison with the microbial utilization of 3,3'-dithiodipropionic acid and 3,3'-thiodipropionic acid. | Wübbeler, JH., et al. 2010. Microbiology (Reading). 156: 1221-1233. PMID: 19959574
- Investigations on the microbial catabolism of the organic sulfur compounds TDP and DTDP in Ralstonia eutropha H16 employing DNA microarrays. | Peplinski, K., et al. 2010. Appl Microbiol Biotechnol. 88: 1145-59. PMID: 20924576
- Polythioester synthesis in Ralstonia eutropha H16: novel insights into 3,3'-thiodipropionic acid and 3,3'-dithiodipropionic acid catabolism. | Doberstein, C., et al. 2014. J Biotechnol. 184: 187-98. PMID: 24953213
- The genome of Variovorax paradoxus strain TBEA6 provides new understandings for the catabolism of 3,3'-thiodipropionic acid and hence the production of polythioesters. | Wübbeler, JH., et al. 2015. J Biotechnol. 209: 85-95. PMID: 26073999
- The NADH:flavin oxidoreductase Nox from Rhodococcus erythropolis MI2 is the key enzyme of 4,4'-dithiodibutyric acid degradation. | Khairy, H., et al. 2016. Lett Appl Microbiol. 63: 434-441. PMID: 27564089
- The unexpected function of a Flavin-dependent oxidoreductase from Variovorax paradoxus TBEA6. | Meinert, C., et al. 2018. FEMS Microbiol Lett. 365: PMID: 29351603
- The catabolism of 3,3'-thiodipropionic acid in Variovorax paradoxus strain TBEA6: A proteomic analysis. | Heine, V., et al. 2019. PLoS One. 14: e0211876. PMID: 30742653
- 3,3'-Thiodipropionic acid (TDP), a possible precursor for the synthesis of polythioesters: identification of TDP transport proteins in Variovorax paradoxus TBEA6. | Reddy, MV. and Steinbüchel, A. 2021. Appl Microbiol Biotechnol. 105: 3733-3743. PMID: 33900422
- Unveiling steps of the TDP degradation pathway in Variovorax paradoxus TBEA6. | Schäfer, L., et al. 2022. Enzyme Microb Technol. 160: 110095. PMID: 35810625
- Combined effects of butylated hydroxyanisole and other antioxidants in induction of forestomach lesions in rats. | Hirose, M., et al. 1986. Cancer Lett. 30: 169-74. PMID: 3955538
- Enhanced recovery of injured Escherichia coli by compounds that degrade hydrogen peroxide or block its formation. | McDonald, LC., et al. 1983. Appl Environ Microbiol. 45: 360-5. PMID: 6338823
- Systematic identification of antioxidants in lards, shortenings, and vegetable oils by thin layer chromatography. | Van Peteghem, CH. and Dekeyser, DA. 1981. J Assoc Off Anal Chem. 64: 1331-5. PMID: 7309653
- Synthesis of water-soluble, nonimmunogenic polyamide cross-linking agents. | Hai, TT., et al. 1998. Bioconjug Chem. 9: 645-54. PMID: 9815156
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
3,3′-Thiodipropionic acid, 250 g | sc-356780 | 250 g | $37.00 | |||
3,3′-Thiodipropionic acid, 1 kg | sc-356780A | 1 kg | $119.00 |
