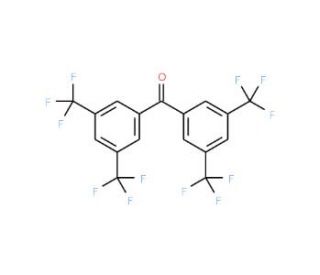
3,3′,5,5′-Tetrakis(trifluoromethyl)benzophenone (CAS 175136-66-0)
QUICK LINKS
3,3′,5,5′-Tetrakis(trifluoromethyl)benzophenone, identified by CAS number 175136-66-0, is a multifunctional organic compound characterized by its robust molecular structure which incorporates four trifluoromethyl groups attached symmetrically to a benzophenone backbone. This chemical is primarily notable for its role in various research domains, particularly in the synthesis of advanced materials and in the study of chemical interactions involving pi-electron systems. The presence of the trifluoromethyl groups significantly enhances the electron-withdrawing capacity of the compound, which in turn influences its reactivity and stability, making it a valuable agent in the creation of polymers and coatings that require enhanced durability against environmental factors. Its application in materials science research often revolves around its ability to act as a building block for the development of high-performance organic compounds. Furthermore, 3,3′,5,5′-Tetrakis(trifluoromethyl)benzophenone is utilized in photochemistry studies where its ability to absorb ultraviolet light is exploited to study the kinetics and mechanisms of photo-induced chemical reactions. This compound, therefore, not only serves as a crucial component in the synthesis of complex molecules but also provides extensive utility in exploring fundamental chemical processes and interactions, enhancing our understanding of molecular behavior under various conditions.
3,3′,5,5′-Tetrakis(trifluoromethyl)benzophenone (CAS 175136-66-0) References
- Metathesis reactions of a manganese borylene complex with polar heteroatom-carbon double bonds: a pathway to previously inaccessible carbene complexes. | Bauer, J., et al. 2013. J Am Chem Soc. 135: 8726-34. PMID: 23692498
- Substituent effect on the photoreduction kinetics of benzophenone. | Demeter, A., et al. 2013. J Phys Chem A. 117: 10196-210. PMID: 23980872
- Highly mesoporous metal-organic framework assembled in a switchable solvent. | Peng, L., et al. 2014. Nat Commun. 5: 4465. PMID: 25047059
- Li vs Na: Divergent Reaction Patterns between Organolithium and Organosodium Complexes and Ligand-Catalyzed Ketone/Aldehyde Methylenation. | Davison, N., et al. 2023. J Am Chem Soc. 145: 6562-6576. PMID: 36890641
- Organocatalytic Michael Addition of Unactivated α-Branched Nitroalkanes to Afford Optically Active Tertiary Nitrocompounds. | Lorea, B., et al. 2023. Org Lett. 25: 8590-8595. PMID: 38009850
- Synthesis, Structures, Dynamics, and Olefin Polymerization Behavior of Group 4 Metal (pyCAr2O) 2M (NR2) 2 Complexes Containing Bidentate Pyridine− Alkoxide Ancillary Ligands. | Kim, Il, et al. 1997. Organometallics. 16.15: 3314-3323.
- Metal-assisted amination with oxime derivatives. | Narasaka, Koichi. 2002. Pure and applied chemistry. 74.1: 143-149.
- Decarbonylation of aryl ketones mediated by bulky cyclopentadienylrhodium bis (ethylene) complexes. | Daugulis, Olafs and Maurice Brookhart. 2004. Organometallics. 23.3: 527-534.
- New C 1-symmetric Ph2C-bridged multisubstituted ansa-zirconocenes for highly isospecific propylene polymerization: Synthetic approach via activated fulvenes. | Kirillov, Evgeny, et al. 2010. Organometallics. 29.21: 5073-5082.
- Base-free aerobic oxidation of alcohols over copper-based complex under ambient condition. | Mei, Qingqing, et al. 2018. ACS sustainable chemistry & engineering. 6.2: 2362-2369.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
3,3′,5,5′-Tetrakis(trifluoromethyl)benzophenone, 1 g | sc-256574 | 1 g | $54.00 |
