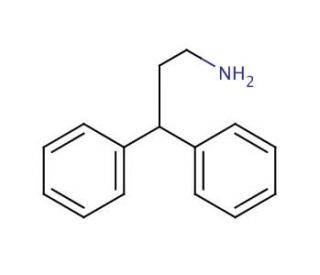
3,3-Diphenylpropylamine (CAS 5586-73-2)
QUICK LINKS
3,3-Diphenylpropylamine functions as a chemical intermediate. It acts as a building block in the synthesis of various organic compounds, contributing to the creation of new materials and substances. At the molecular level, 3,3-Diphenylpropylamine participates in reactions that lead to the formation of complex structures, enabling the production of novel compounds with potential applications in diverse fields. Its mechanism of action involves participating in chemical reactions as a reactant or reagent, facilitating the transformation of precursor molecules into more complex products. 3,3-Diphenylpropylamine plays a role in the development of new chemical entities and materials, contributing to the advancement of scientific knowledge and innovation in various industries.
3,3-Diphenylpropylamine (CAS 5586-73-2) References
- Determination of the D- and L-enantiomers of modafinil in human plasma utilizing liquid-liquid extraction and high-performance liquid chromatography. | Gorman, SH. 1999. J Chromatogr B Biomed Sci Appl. 730: 1-7. PMID: 10437666
- NPS 1506, a novel NMDA receptor antagonist and neuroprotectant. Review of preclinical and clinical studies. | Mueller, AL., et al. 1999. Ann N Y Acad Sci. 890: 450-7. PMID: 10668449
- A novel N-methyl-D-aspartate receptor open channel blocker with in vivo neuroprotectant activity. | Planells-Cases, R., et al. 2002. J Pharmacol Exp Ther. 302: 163-73. PMID: 12065713
- Chiral analysis of d- and l-modafinil in human serum: application to human pharmacokinetic studies. | Donovan, JL., et al. 2003. Ther Drug Monit. 25: 197-202. PMID: 12657914
- 3-Chloro,4-methoxyfendiline is a potent GABA(B) receptor potentiator in rat neocortical slices. | Ong, J., et al. 2005. Eur J Pharmacol. 507: 35-42. PMID: 15659292
- Identification from a positional scanning peptoid library of in vivo active compounds that neutralize bacterial endotoxins. | Mora, P., et al. 2005. J Med Chem. 48: 1265-8. PMID: 15715495
- Anchoring dyes into multidimensional large-pore zeolites: a prospective use as chromogenic sensing materials. | Comes, M., et al. 2006. Chemistry. 12: 2162-70. PMID: 16374890
- Novel sterically hindered cannabinoid CB1 receptor ligands. | Urbani, P., et al. 2008. Bioorg Med Chem. 16: 7510-5. PMID: 18579386
- Identification and characterization of urinary prenylamine metabolites by means of liquid chromatography-tandem mass spectrometry. | Beuck, S., et al. 2012. Drug Test Anal. 4: 701-16. PMID: 22786790
- Chemically Addressable Perovskite Nanocrystals for Light-Emitting Applications. | Sun, H., et al. 2017. Adv Mater. 29: PMID: 28692786
- Design, Synthesis, and Biological Evaluation of 1-Benzylamino-2-hydroxyalkyl Derivatives as New Potential Disease-Modifying Multifunctional Anti-Alzheimer's Agents. | Panek, D., et al. 2018. ACS Chem Neurosci. 9: 1074-1094. PMID: 29345897
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
3,3-Diphenylpropylamine, 1 g | sc-231945A | 1 g | $42.00 | |||
3,3-Diphenylpropylamine, 5 g | sc-231945B | 5 g | $54.00 | |||
3,3-Diphenylpropylamine, 25 g | sc-231945 | 25 g | $124.00 | |||
3,3-Diphenylpropylamine, 100 g | sc-231945C | 100 g | $364.00 |
