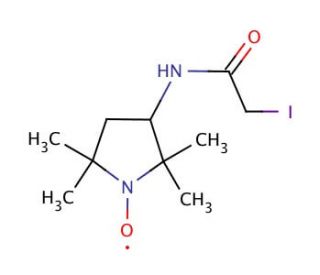
3-(2-Iodoacetamido)-PROXYL (CAS 27048-01-7)
QUICK LINKS
3-(2-Iodoacetamido)-PROXYL is a spin label compound utilized in the research field of electron paramagnetic resonance (EPR) spectroscopy, particularly in the study of protein structure and dynamics. As a spin label, it is covalently attached to proteins or other biomolecules through its iodoacetamido functional group, which reacts with thiol groups in cysteine residues. This labeling allows for the measurement of distances within proteins, protein-protein interactions, and conformational changes upon ligand binding or during enzymatic reactions. In addition, 3-(2-Iodoacetamido)-PROXYL is used to investigate the properties of membranes and the positioning and movement of proteins within lipid bilayers. By measuring the EPR spectra of labeled molecules, researchers can gain insights into the local microenvironment and molecular motions, which are critical for understanding the function of biological macromolecules.
3-(2-Iodoacetamido)-PROXYL (CAS 27048-01-7) References
- NMR study of the sites of human hemoglobin acetylated by aspirin. | Xu, AS., et al. 1999. Biochim Biophys Acta. 1432: 333-49. PMID: 10407155
- Measurement of large distances in biomolecules using double-quantum filtered refocused electron spin-echoes. | Borbat, PP., et al. 2004. J Am Chem Soc. 126: 7746-7. PMID: 15212500
- Distance measurements on spin-labelled biomacromolecules by pulsed electron paramagnetic resonance. | Jeschke, G. and Polyhach, Y. 2007. Phys Chem Chem Phys. 9: 1895-910. PMID: 17431518
- Refolding of the integral membrane protein light-harvesting complex II monitored by pulse EPR. | Dockter, C., et al. 2009. Proc Natl Acad Sci U S A. 106: 18485-90. PMID: 19833872
- Bacterial collagen-binding domain targets undertwisted regions of collagen. | Philominathan, ST., et al. 2012. Protein Sci. 21: 1554-65. PMID: 22898990
- Preparation of cysteine-34-nitroxide spin labeled human α₁-microglobulin. | Nalepa, AI., et al. 2013. Protein Expr Purif. 88: 33-40. PMID: 23201281
- Role of the C-terminal domain in the structure and function of tetrameric sodium channels. | Bagnéris, C., et al. 2013. Nat Commun. 4: 2465. PMID: 24051986
- Functional role of the flexible N-terminal extension of FKBP38 in catalysis. | Kang, C., et al. 2013. Sci Rep. 3: 2985. PMID: 24145868
- EPR-aided approach for solution structure determination of large RNAs or protein-RNA complexes. | Duss, O., et al. 2014. Nat Commun. 5: 3669. PMID: 24828280
- A fully enzymatic method for site-directed spin labeling of long RNA. | Lebars, I., et al. 2014. Nucleic Acids Res. 42: e117. PMID: 24981512
- Activation of the A2A adenosine G-protein-coupled receptor by conformational selection. | Ye, L., et al. 2016. Nature. 533: 265-8. PMID: 27144352
- Spinach leaf ribulose-5-phosphate kinase: examination of sulfhydryls by chemical modification and spin-labeling. | Krieger, TJ. and Miziorko, HM. 1987. Arch Biochem Biophys. 256: 362-71. PMID: 3038027
- Site-Specific Spin Labeling of RNA for NMR and EPR Structural Studies. | Vileno, B. and Lebars, I. 2020. Methods Mol Biol. 2113: 217-235. PMID: 32006317
- Strategies for Covalent Labeling of Long RNAs. | Depmeier, H., et al. 2021. Chembiochem. 22: 2826-2847. PMID: 34043861
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
3-(2-Iodoacetamido)-PROXYL, 10 mg | sc-209450 | 10 mg | $275.00 |
