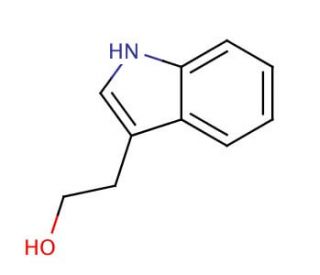
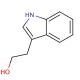
Molecular structure of 3-(2-Hydroxyethyl)indole, CAS Number: 526-55-6
3-(2-Hydroxyethyl)indole (CAS 526-55-6)
Application:
3-(2-Hydroxyethyl)indole is an indole for the production of compounds used in biological research
CAS Number:
526-55-6
Purity:
≥98%
Molecular Weight:
161.20
Molecular Formula:
C10H11NO
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
3-(2-Hydroxyethyl)indole is a substrate in various enzymatic reactions. It acts as a precursor in the synthesis of indole derivatives, which are important in the development of agrochemicals. 3-(2-Hydroxyethyl)Indole undergoes specific chemical transformations, leading to the formation of new molecular structures with potential biological activities. At the molecular level, 3-(2-Hydroxyethyl)indole interacts with enzymes and other chemical reagents to facilitate the production of diverse indole-based compounds.
3-(2-Hydroxyethyl)indole (CAS 526-55-6) References
- The pyruvate kinase of Stigmatella aurantiaca is an indole binding protein and essential for development. | Stamm, I., et al. 2005. Mol Microbiol. 56: 1386-95. PMID: 15882428
- N-carboxamido-staurosporine and selina-4(14),7(11)-diene-8,9-diol, new metabolites from a marine Streptomyces sp. | Wu, SJ., et al. 2006. J Antibiot (Tokyo). 59: 331-7. PMID: 16915816
- Structure-activity relationships of 2,N(6),5'-substituted adenosine derivatives with potent activity at the A2B adenosine receptor. | Adachi, H., et al. 2007. J Med Chem. 50: 1810-27. PMID: 17378544
- The L293 residue in transmembrane domain 2 of the 5-HT3A receptor is a molecular determinant of allosteric modulation by 5-hydroxyindole. | Hu, XQ. and Lovinger, DM. 2008. Neuropharmacology. 54: 1153-65. PMID: 18436267
- Which agonist properties are important for the activation of 5-HT3A receptors? | Meiboom, MF., et al. 2013. Biochim Biophys Acta. 1828: 2564-73. PMID: 23792067
- Polyphenols and Tryptophan Metabolites Activate the Aryl Hydrocarbon Receptor in an in vitro Model of Colonic Fermentation. | Koper, JEB., et al. 2019. Mol Nutr Food Res. 63: e1800722. PMID: 30443985
- XszenFHal, a novel tryptophan 5-halogenase from Xenorhabdus szentirmaii. | Domergue, J., et al. 2019. AMB Express. 9: 175. PMID: 31673806
- Bioconversion of Biologically Active Indole Derivatives with Indole-3-Acetic Acid-Degrading Enzymes from Caballeronia glathei DSM50014. | Sadauskas, M., et al. 2020. Biomolecules. 10: PMID: 32344740
- Antibiofilm and Antivirulence Properties of Indoles Against Serratia marcescens. | Sethupathy, S., et al. 2020. Front Microbiol. 11: 584812. PMID: 33193228
- Metabolomic Expression of Laryngeal and Hindlimb Muscles in Adult versus Senescent Rats. | Shembel, AC., et al. 2022. Ann Otol Rhinol Laryngol. 131: 259-267. PMID: 34041924
- Liquid chromatography-tandem mass spectrometry analysis for identification and quantification of antimicrobial compounds in distillery wastewater. | Hou, W., et al. 2021. MethodsX. 8: 101470. PMID: 34430343
- Characteristics of Serum Metabolites and Gut Microbiota in Diabetic Kidney Disease. | Zhang, B., et al. 2022. Front Pharmacol. 13: 872988. PMID: 35548353
- Identifying potential biomarkers and therapeutic targets for dogs with sepsis using metabolomics and lipidomics analyses. | Montague, B., et al. 2022. PLoS One. 17: e0271137. PMID: 35802586
- Comparative Metagenomics and Metabolomes Reveals Abnormal Metabolism Activity Is Associated with Gut Microbiota in Alzheimer's Disease Mice. | Sun, P., et al. 2022. Int J Mol Sci. 23: PMID: 36232865
- Screening of Endophytic Bacteria of Leucojum aestivum 'Gravety Giant' as a Potential Source of Alkaloids and as Antagonist to Some Plant Fungal Pathogens. | Munakata, Y., et al. 2022. Microorganisms. 10: PMID: 36296365
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
3-(2-Hydroxyethyl)indole, 1 g | sc-256394A | 1 g | $47.00 | |||
3-(2-Hydroxyethyl)indole, 5 g | sc-256394 | 5 g | $104.00 | |||
3-(2-Hydroxyethyl)indole, 25 g | sc-256394B | 25 g | $349.00 | |||
3-(2-Hydroxyethyl)indole, 100 g | sc-256394C | 100 g | $883.00 |
