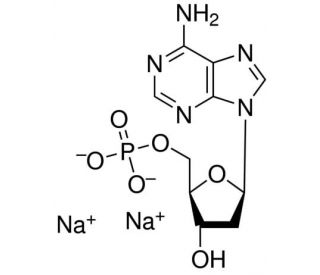
2′-Deoxyadenosine 5′-monophosphate sodium salt (CAS 2922-74-9)
See product citations (1)
QUICK LINKS
2′-Deoxyadenosine 5′-monophosphate sodium salt, often abbreviated as dAMP, is a nucleotide derivative that is pivotal in the realms of molecular biology and genetic research. This compound, specifically the sodium salt form, enhances solubility and stability, making it particularly useful in various laboratory applications. 2′-Deoxyadenosine 5′-monophosphate sodium salt serves as a fundamental building block in the synthesis of DNA, where it incorporates into the growing DNA strand during the replication and transcription processes. Its structure, lacking an oxygen atom at the 2′ position of the ribose sugar, distinguishes it from ribonucleotides and is critical for DNA′s stability and functional properties. In research, 2′-Deoxyadenosine 5′-monophosphate sodium salt is extensively used in studies involving DNA polymerase reactions, sequencing, and nucleic acid amplification techniques. Its role is in understanding the mechanisms of genetic expression, mutation, and repair, thereby providing insights into the fundamental processes that govern cellular function and heredity.
2′-Deoxyadenosine 5′-monophosphate sodium salt (CAS 2922-74-9) References
- A simple copper(II)-L-histidine system for efficient hydrolytic cleavage of DNA. | Ren, R., et al. 2000. Inorg Chem. 39: 5454-63. PMID: 11154560
- Accurate nucleic acid concentrations by nuclear magnetic resonance. | Cavaluzzi, MJ., et al. 2002. Anal Biochem. 308: 373-80. PMID: 12419352
- Nitrogen chemical structure in DNA and related molecules by X-ray absorption spectroscopy. | Kirtley, SM., et al. 1992. Biochim Biophys Acta. 1132: 249-54. PMID: 1420304
- Improved detection limit for a direct determination of 8-hydroxy-2'-deoxyguanosine in untreated urine samples by capillary electrophoresis with optical detection. | Tůma, P., et al. 2004. J Chromatogr B Analyt Technol Biomed Life Sci. 813: 255-61. PMID: 15556541
- Determination of apurinic/apyrimidinic lesions in DNA with high-performance liquid chromatography and tandem mass spectrometry. | Roberts, KP., et al. 2006. Chem Res Toxicol. 19: 300-9. PMID: 16485907
- Dietary nucleotides and human immune cells. II. Modulation of PBMC growth and cytokine secretion. | Holen, E., et al. 2006. Nutrition. 22: 90-6. PMID: 16615178
- Non-enzymatic transfer of sequence information under plausible prebiotic conditions. | Olasagasti, F., et al. 2011. Biochimie. 93: 556-61. PMID: 21130835
- Enhancing lipophilicity as a strategy to overcome resistance against platinum complexes? | Buss, I., et al. 2011. J Inorg Biochem. 105: 709-17. PMID: 21450275
- Understanding mechanisms of pressure-assisted electrokinetic injection: application to analysis of bromate, arsenic and selenium species in drinking water by capillary electrophoresis-mass spectrometry. | Zhang, H., et al. 2011. J Chromatogr A. 1218: 3095-104. PMID: 21489539
- Characterization of DNA adducts derived from (+/-)-trans-3,4-dihydroxy-anti-1,2-epoxy-1,2,3,4- tetrahydrodibenz[a,j]anthracene and (+/-)-7-methyl-trans-3,4-dihydroxy- anti-1,2-epoxy-1,2,3,4-tetrahydrodibenz[a,j]anthracene. | Nair, RV., et al. 1989. Chem Res Toxicol. 2: 341-8. PMID: 2519825
- The effect of fluoro substituents on reactivity of 7-methylbenz[a]anthracene diol epoxides. | Baer-Dubowska, W., et al. 1996. Chem Res Toxicol. 9: 722-8. PMID: 8831816
- In-water reactivity of nucleosides and nucleotides: one-step preparation and biological evaluation of novel ferrocenyl-derivatives | M de Champdoré, G Di Fabio, A Messere. 2004. Tetrahedron. 60(31): 6555-6563.
- DNA sequences-mediated fine-tuning of nanostructures and their plasmonic properties over gold nanorods | H Zhao, T Jiang, L Yi, L Tang. 2021. Optik. 228: 166137.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2′-Deoxyadenosine 5′-monophosphate sodium salt, 250 mg | sc-338742 | 250 mg | $151.00 | |||
2′-Deoxyadenosine 5′-monophosphate sodium salt, 500 mg | sc-338742A | 500 mg | $154.00 | |||
2′-Deoxyadenosine 5′-monophosphate sodium salt, 1 g | sc-338742B | 1 g | $317.00 | |||
2′-Deoxyadenosine 5′-monophosphate sodium salt, 5 g | sc-338742C | 5 g | $1462.00 |
