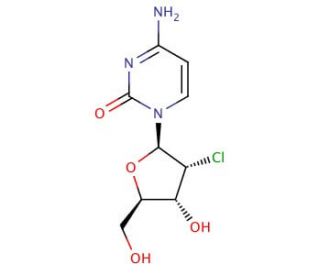
2′-Chloro-2′-deoxycytidine (CAS 10212-19-8)
QUICK LINKS
2′-Chloro-2′-deoxycytidine is a nucleoside analog used extensively in biochemical and molecular biology research, particularly in studies of DNA synthesis and repair mechanisms. Its mechanism of action involves incorporation into DNA during replication, where it can induce chain termination or cause mutations due to its altered structure. This analog is instrumental in understanding the fidelity and processivity of DNA polymerases, enzymes responsible for synthesizing new DNA strands. Researchers use 2′-chloro-2′-deoxycytidine to investigate the effects of nucleotide analogs on DNA replication and to study the mechanisms of nucleotide excision repair, a critical cellular process that removes damaged nucleotides from DNA. Additionally, it is utilized in research exploring the replication fidelity of various DNA polymerases, including those involved in viral replication, providing insights into antiviral strategies. The compound′s ability to induce specific types of DNA damage makes it a valuable tool in studying mutagenesis and the cellular response to DNA damage, including the activation of DNA damage checkpoints and repair pathways. Recent studies have focused on its application in understanding the mechanisms of drug resistance in cancer cells, where altered DNA repair capabilities can impact the effectiveness of nucleoside analogs. Overall, 2′-chloro-2′-deoxycytidine remains a crucial compound in the study of DNA synthesis, repair, and mutagenesis.
2′-Chloro-2′-deoxycytidine (CAS 10212-19-8) References
- A packing-density metric for exploring the interior of folded RNA molecules. | Schwans, JP., et al. 2004. Angew Chem Int Ed Engl. 43: 3033-7. PMID: 15188472
- Hydrolysis in 2'-chloro-2'-deoxy nucleosides, -nucleotides and -polynucleotides to arabinose-derivatives. | Hobbs, J. and Eckstein, F. 1975. Nucleic Acids Res. 2: 1987-94. PMID: 174061
- Ribonucleotide Reductases: Structure, Chemistry, and Metabolism Suggest New Therapeutic Targets. | Greene, BL., et al. 2020. Annu Rev Biochem. 89: 45-75. PMID: 32569524
- Synthesis and properties of poly 2'-fluoro-2'-deoxyuridylic acid. | Janik, B., et al. 1972. Biochem Biophys Res Commun. 46: 1153-60. PMID: 4334969
- Studies on the Vilsmeier-Haack reaction. IV. Convenient synthesis of 2,2'-anhydro-1- -D-arabinofuranosylcytosine (2,2'-cyclocytidine) and its derivatives. | Kikugawa, K. and Ichino, M. 1972. J Org Chem. 37: 284-8. PMID: 5013349
- Nucleosides. XXXIX. 2'-deoxy-2'-fluorocytidine, 1-beta-D-arabinofuranosyl-2-amino-1,4(2H)-4-iminopyrimidine, and related derivatives. | Doerr, IL. and Fox, JJ. 1967. J Org Chem. 32: 1462-71. PMID: 6041430
- Characterization of the active site of ribonucleotide reductase of Escherichia coli, bacteriophage T4 and mammalian cells by inhibition studies with hydroxyurea analogues. | Larsen, IK., et al. 1982. Eur J Biochem. 125: 75-81. PMID: 7049700
- Interactions of 2'-modified azido- and haloanalogs of deoxycytidine 5'-triphosphate with the anaerobic ribonucleotide reductase of Escherichia coli. | Eliasson, R., et al. 1994. J Biol Chem. 269: 26116-20. PMID: 7929323
- Mechanism of inactivation of Escherichia coli and Lactobacillus leichmannii ribonucleotide reductases by 2'-chloro-2'-deoxynucleotides: evidence for generation of 2-methylene-3 (2H)-furanone[J]. | Harris G, Ator M, Stubbe J. 1984,. Biochemistry,. 23(22):: 5214-5225.
- Design, Synthesis, and Antitumor Activity of an Inhibitor of Ribonucleotide Reductase[M]//Chemical and Structural Approaches to Rational Drug Design. | McCarthy J R, Sunkara P S. 2020. CRC Press,.: 3-: 34.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2′-Chloro-2′-deoxycytidine, 1 g | sc-394096 | 1 g | $3125.00 | |||
2′-Chloro-2′-deoxycytidine, 5 g | sc-394096A | 5 g | $12340.00 |
